Korean J Radiol.
2018 Jun;19(3):431-442. 10.3348/kjr.2018.19.3.431.
Altered White Matter Integrity in Human Immunodeficiency Virus-Associated Neurocognitive Disorder: A Tract-Based Spatial Statistics Study
- Affiliations
-
- 1Department of Radiology, Soonchunhyang University Cheonan Hospital, Cheonan 31151, Korea.
- 2Department of Radiology, College of Medicine, The Catholic University of Korea, Seoul 06591, Korea. nyshin@catholic.ac.kr
- 3Department of Internal Medicine and AIDS Research Institute, Yonsei University College of Medicine, Seoul 03722, Korea.
- 4Department of Radiology, Research Institute of Radiological Science, Yonsei University College of Medicine, Seoul 03722, Korea.
- KMID: 2410814
- DOI: http://doi.org/10.3348/kjr.2018.19.3.431
Abstract
OBJECTIVE
Human immunodeficiency virus (HIV) infection has been known to damage the microstructural integrity of white matter (WM). However, only a few studies have assessed the brain regions in HIV-associated neurocognitive disorders (HAND) with diffusion tensor imaging (DTI). Therefore, we sought to compare the DTI data between HIV patients with and without HAND using tract-based spatial statistics (TBSS).
MATERIALS AND METHODS
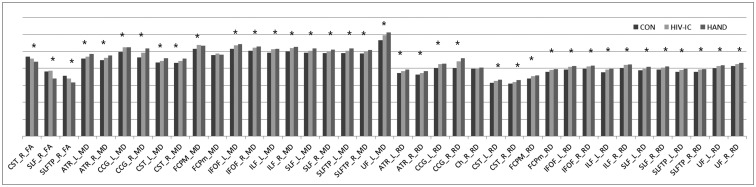
Twenty-two HIV-infected patients (10 with HAND and 12 without HAND) and 11 healthy controls (HC) were enrolled in this study. A whole-brain analysis of fractional anisotropy (FA), mean diffusivity (MD), radial diffusivity (RD), and axial diffusivity was performed with TBSS and a subsequent 20 tract-specific region-of-interest (ROI)-based analysis to localize and compare altered WM integrity in all group contrasts.
RESULTS
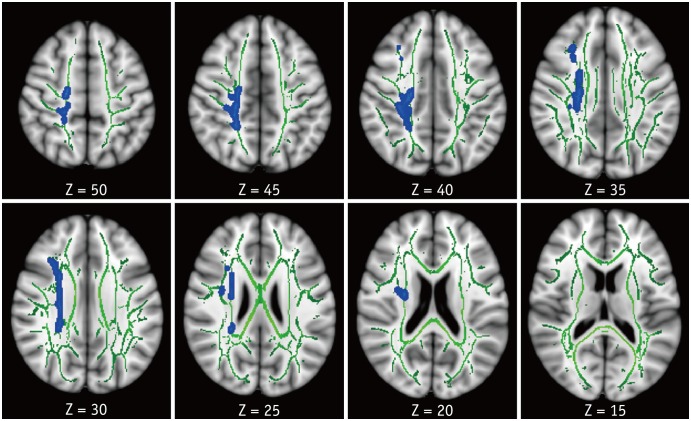
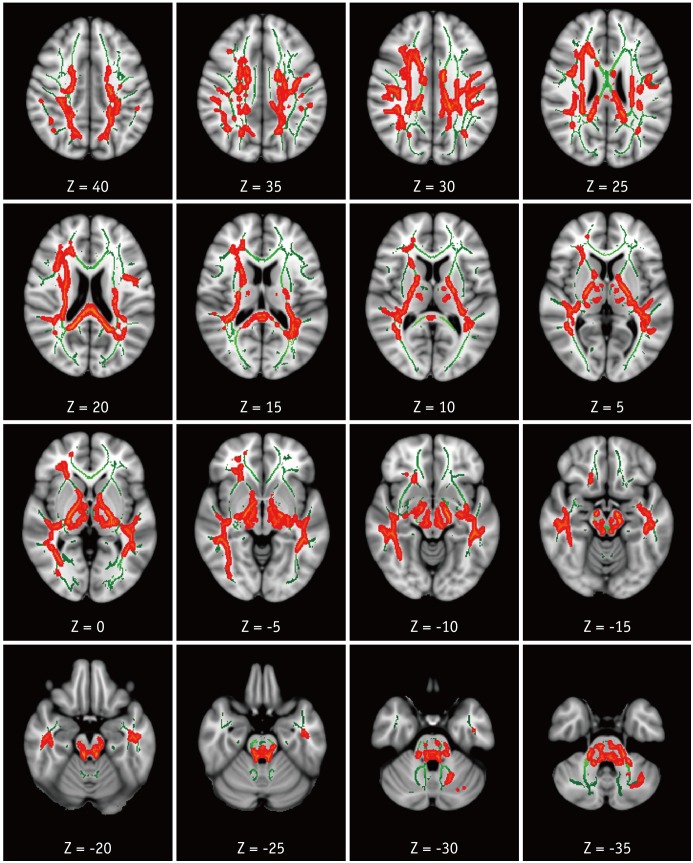
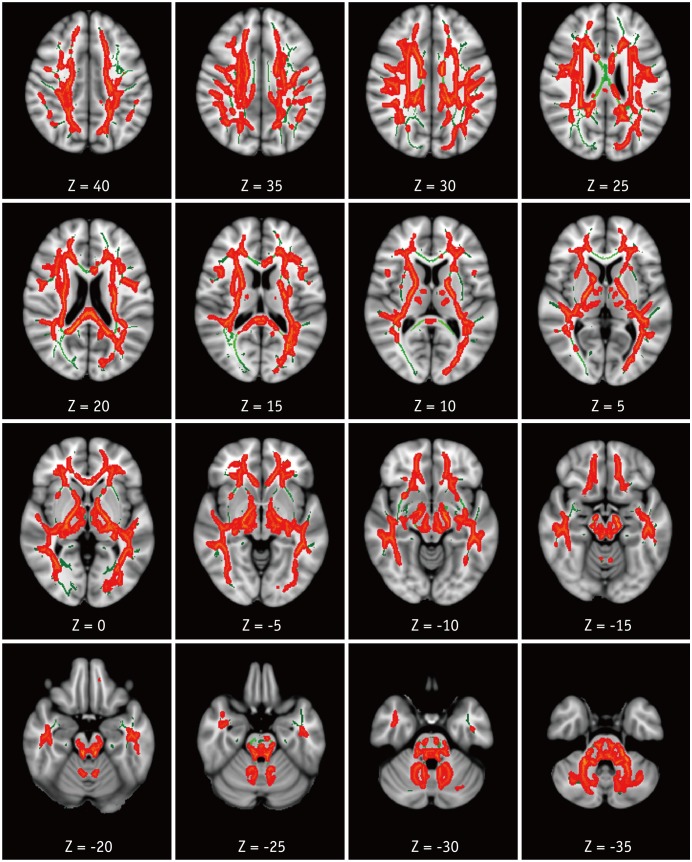
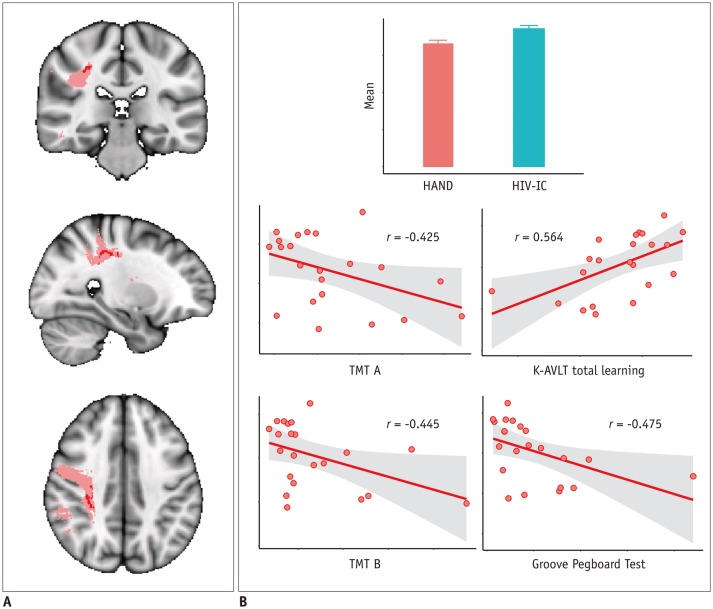
Compared with HC, patients with HAND showed decreased FA in the right frontoparietal WM including the upper corticospinal tract (CST) and increased MD and RD in the bilateral frontoparietal WM, corpus callosum, bilateral CSTs and bilateral cerebellar peduncles. The DTI values did not significantly differ between HIV patients with and without HAND or between HIV patients without HAND and HC. In the ROI-based analysis, decreased FA was observed in the right superior longitudinal fasciculus and was significantly correlated with decreased information processing speed, memory, executive function, and fine motor function in HIV patients.
CONCLUSION
These results suggest that altered integrity of the frontoparietal WM contributes to cognitive dysfunction in HIV patients.
Keyword
MeSH Terms
Figure
Reference
-
1. Holt JL, Kraft-Terry SD, Chang L. Neuroimaging studies of the aging HIV-1-infected brain. J Neurovirol. 2012; 18:291–302. PMID: 22653528.
Article2. Schouten J, Cinque P, Gisslen M, Reiss P, Portegies P. HIV-1 infection and cognitive impairment in the cART era: a review. AIDS. 2011; 25:561–575. PMID: 21160410.
Article3. Dore GJ, McDonald A, Li Y, Kaldor JM, Brew BJ. National HIV Surveillance Committee. Marked improvement in survival following AIDS dementia complex in the era of highly active antiretroviral therapy. AIDS. 2003; 17:1539–1545. PMID: 12824792.
Article4. Ku NS, Lee Y, Ahn JY, Song JE, Kim MH, Kim SB, et al. HIV-associated neurocognitive disorder in HIV-infected Koreans: the Korean NeuroAIDS Project. HIV Med. 2014; 15:470–477. PMID: 24580888.
Article5. Chernoff RA, Martin DJ, Schrock DA, Huy MP. Neuropsychological functioning as a predictor of employment activity in a longitudinal study of HIV-infected adults contemplating workforce reentry. J Int Neuropsychol Soc. 2010; 16:38–48. PMID: 19765357.
Article6. Antinori A, Arendt G, Becker JT, Brew BJ, Byrd DA, Cherner M, et al. Updated research nosology for HIV-associated neurocognitive disorders. Neurology. 2007; 69:1789–1799. PMID: 17914061.
Article7. Hill-Briggs F, Dial JG, Morere DA, Joyce A. Neuropsychological assessment of persons with physical disability, visual impairment or blindness, and hearing impairment or deafness. Arch Clin Neuropsychol. 2007; 22:389–404. PMID: 17303374.8. Ostrosky-Solis F, Ardila A, Rosselli M, Lopez-Arango G, Uriel-Mendoza V. Neuropsychological test performance in illiterate subjects. Arch Clin Neuropsychol. 1998; 13:645–660. PMID: 14590626.
Article9. Masters MC, Ances BM. Role of neuroimaging in HIV-associated neurocognitive disorders. Semin Neurol. 2014; 34:89–102. PMID: 24715492.
Article10. Karampekios S, Hesselink J. Cerebral infections. Eur Radiol. 2005; 15:485–493. PMID: 15627191.
Article11. Sundgren PC, Dong Q, Gómez-Hassan D, Mukherji SK, Maly P, Welsh R. Diffusion tensor imaging of the brain: review of clinical applications. Neuroradiology. 2004; 46:339–350. PMID: 15103435.
Article12. Dore GJ, Correll PK, Li Y, Kaldor JM, Cooper DA, Brew BJ. Changes to AIDS dementia complex in the era of highly active antiretroviral therapy. AIDS. 1999; 13:1249–1253. PMID: 10416530.
Article13. Heaton RK, Chelune GJ, Talley JL, Kay GG, Curtiss G. Wisconsin card sorting test manual: revised and expanded. Odessa, FL: Psychological Assessment Resources;1993. p. 92–193.14. Kim HK. Handbook of Rey-Kim memory assessment. Daegu: Neuropsychology Press;1999. p. 130–148.15. Kim MK, Hyun MH. Relationships between Trail Making Test (A, B, B-A. B/A) scores and ape, education, comparison of performance head injury patient and psychiatric patient. Korean J Clin Psychol. 2004; 23:353–366.16. Lee T. Normative values for the Grooved Pegboard Test in adult. Phys Ther Korea. 2001; 8:87–94.17. Yeom TH, Park YS, Oh KJ, Kim JK, Lee YH. K-WAIS manual. Seoul: Korea Guidance;1992. p. 105–119.18. Smith SM, Jenkinson M, Johansen-Berg H, Rueckert D, Nichols TE, Mackay CE, et al. Tract-based spatial statistics: voxelwise analysis of multi-subject diffusion data. Neuroimage. 2006; 31:1487–1505. PMID: 16624579.
Article19. Purohit V, Rapaka R, Frankenheim J, Avila A, Sorensen R, Rutter J. National Institute on Drug Abuse symposium report: drugs of abuse, dopamine, and HIV-associated neurocognitive disorders/HIV-associated dementia. J Neurovirol. 2013; 19:119–122. PMID: 23456951.
Article20. Mediouni S, Marcondes MC, Miller C, McLaughlin JP, Valente ST. The cross-talk of HIV-1 Tat and methamphetamine in HIV-associated neurocognitive disorders. Front Microbiol. 2015; 6:1164. PMID: 26557111.
Article21. Lentz MR, Kim WK, Kim H, Soulas C, Lee V, Venna N, et al. Alterations in brain metabolism during the first year of HIV infection. J Neurovirol. 2011; 17:220–229. PMID: 21494901.
Article22. Valcour V, Chalermchai T, Sailasuta N, Marovich M, Lerdlum S, Suttichom D, et al. Central nervous system viral invasion and inflammation during acute HIV infection. J Infect Dis. 2012; 206:275–282. PMID: 22551810.
Article23. Anthony IC, Bell JE. The neuropathology of HIV/AIDS. Int Rev Psychiatry. 2008; 20:15–24. PMID: 18240059.
Article24. Reiche EMV, Moritmoto HK, de Almeida ERD, Oliveira SR, Kallaur AP, Simão ANC. Oxidative stress and human immunodeficiency virus type 1 (HIV-1) infection. In : Dichi I, Breganó JW, Simão ANC, Cecchini R, editors. Role of oxidative stress in chronic diseases. Boca Raton, FL: CRC Press;2014. p. 45–89.25. Barber SC, Shaw PJ. Oxidative stress in ALS: key role in motor neuron injury and therapeutic target. Free Radic Biol Med. 2010; 48:629–641. PMID: 19969067.
Article26. Wang X, Michaelis EK. Selective neuronal vulnerability to oxidative stress in the brain. Front Aging Neurosci. 2010; 2:12. PMID: 20552050.
Article27. Chen Y, An H, Zhu H, Stone T, Smith JK, Hall C, et al. White matter abnormalities revealed by diffusion tensor imaging in non-demented and demented HIV+ patients. Neuroimage. 2009; 47:1154–1162. PMID: 19376246.
Article28. Corrêa DG, Zimmermann N, Doring TM, Wilner NV, Leite SC, Cabral RF, et al. Diffusion tensor MR imaging of white matter integrity in HIV-positive patients with planning deficit. Neuroradiology. 2015; 57:475–482. PMID: 25604843.
Article29. Feldman HM, Yeatman JD, Lee ES, Barde LH, Gaman-Bean S. Diffusion tensor imaging: a review for pediatric researchers and clinicians. J Dev Behav Pediatr. 2010; 31:346–356. PMID: 20453582.
Article30. Stebbins GT, Smith CA, Bartt RE, Kessler HA, Adeyemi OM, Martin E, et al. HIV-associated alterations in normal-appearing white matter: a voxel-wise diffusion tensor imaging study. J Acquir Immune Defic Syndr. 2007; 46:564–573. PMID: 18193498.31. Wu Y, Storey P, Cohen BA, Epstein LG, Edelman RR, Ragin AB. Diffusion alterations in corpus callosum of patients with HIV. AJNR Am J Neuroradiol. 2006; 27:656–660. PMID: 16552012.32. Zhu T, Zhong J, Hu R, Tivarus M, Ekholm S, Harezlak J, et al. Patterns of white matter injury in HIV infection after partial immune reconstitution: a DTI tract-based spatial statistics study. J Neurovirol. 2013; 19:10–23. PMID: 23179680.
Article33. Budde MD, Xie M, Cross AH, Song SK. Axial diffusivity is the primary correlate of axonal injury in the experimental autoimmune encephalomyelitis spinal cord: a quantitative pixelwise analysis. J Neurosci. 2009; 29:2805–2813. PMID: 19261876.
Article34. Shin NY, Hong J, Choi JY, Lee SK, Lim SM, Yoon U. Retrosplenial cortical thinning as a possible major contributor for cognitive impairment in HIV patients. Eur Radiol. 2017; 27:4721–4729. PMID: 28409354.
Article35. Thompson PM, Dutton RA, Hayashi KM, Toga AW, Lopez OL, Aizenstein HJ, et al. Thinning of the cerebral cortex visualized in HIV/AIDS reflects CD4+ T lymphocyte decline. Proc Natl Acad Sci U S A. 2005; 102:15647–15652. PMID: 16227428.
Article36. Stubbe-Drger B, Deppe M, Mohammadi S, Keller SS, Kugel H, Gregor N, et al. ; German Competence Network HIV/AIDS. Early microstructural white matter changes in patients with HIV: a diffusion tensor imaging study. BMC Neurol. 2012; 12:23. PMID: 22548835.
Article37. Schmahmann JD, Smith EE, Eichler FS, Filley CM. Cerebral white matter: neuroanatomy, clinical neurology, and neurobehavioral correlates. Ann N Y Acad Sci. 2008; 1142:266–309. PMID: 18990132.38. Turken A, Whitfield-Gabrieli S, Bammer R, Baldo JV, Dronkers NF, Gabrieli JD. Cognitive processing speed and the structure of white matter pathways: convergent evidence from normal variation and lesion studies. Neuroimage. 2008; 42:1032–1044. PMID: 18602840.
Article39. Gutiérrez R, Boison D, Heinemann U, Stoffel W. Decompaction of CNS myelin leads to a reduction of the conduction velocity of action potentials in optic nerve. Neurosci Lett. 1995; 195:93–96. PMID: 7478276.
Article40. Tolhurst DJ, Lewis PR. Effect of myelination on the conduction velocity of optic nerve fibres. Ophthalmic Physiol Opt. 1992; 12:241–243. PMID: 1408181.
Article41. Waxman SG. Determinants of conduction velocity in myelinated nerve fibers. Muscle Nerve. 1980; 3:141–150. PMID: 6245357.
Article42. Wilson TW, Heinrichs-Graham E, Robertson KR, Sandkovsky U, O'Neill J, Knott NL, et al. Functional brain abnormalities during finger-tapping in HIV-infected older adults: a magnetoencephalography study. J Neuroimmune Pharmacol. 2013; 8:965–974. PMID: 23749418.
Article43. Chung SJ, Shin JH, Cho KH, Lee Y, Sohn YH, Seong JK, et al. Subcortical shape analysis of progressive mild cognitive impairment in Parkinson's disease. Mov Disord. 2017; 32:1447–1456. PMID: 28737237.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Diffusion Tensor Imaging Findings of White Matter Changes in First Episode Schizophrenia: A Systematic Review
- Evaluation of White Matter Abnormality in Mild Alzheimer Disease and Mild Cognitive Impairment Using Diffusion Tensor Imaging: A Comparison of Tract-Based Spatial Statistics with Voxel-Based Morphometry
- Association Between the C4 Binding Protein Level and White Matter Integrity in Major Depressive Disorder
- Impact of Metabolic Syndrome on the White Matter Integrity in Alzheimer's Disease Patients : Tract-Based Spatial Statistics Study under 3-Tesla MRI
- A Case of Ankylosing Spondylitis in a Patient with Human Immunodeficiency Virus