Ann Lab Med.
2016 Jan;36(1):76-78. 10.3343/alm.2016.36.1.76.
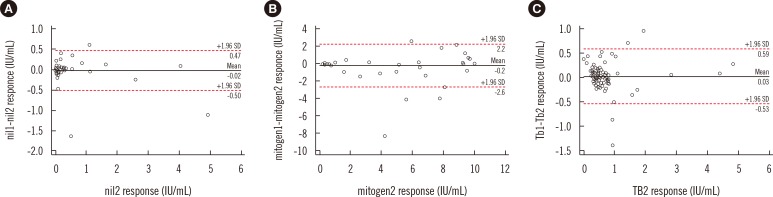
Repeatability of QuantiFERON-TB Gold In-Tube Assay Results Near Cut-Off Points
- Affiliations
-
- 1Department of Laboratory Medicine, Dong-A University College of Medicine, Busan, Korea. progreen@dau.ac.kr
- KMID: 2373504
- DOI: http://doi.org/10.3343/alm.2016.36.1.76
Abstract
- No abstract available.
MeSH Terms
Figure
Reference
-
1. Mazurek GH, Jereb J, Vernon A, LoBue P, Goldberg S, Castro K. IGRA Expert Committee. Centers for Disease Control and Prevention (CDC). Updated guidelines for using Interferon Gamma Release Assays to detect Mycobacterium tuberculosis infection-United States, 2010. MMWR Recomm Rep. 2010; 59:1–25. PMID: 20577159.2. Whitworth HS, Scott M, Connell DW, Dongés B, Lalvani A. IGRAs--the gateway to T cell based TB diagnosis. Methods. 2013; 61:52–62. PMID: 23296020.3. Park JS, Lee JS, Kim MY, Lee CH, Yoon HI, Lee SM, et al. Monthly follow-ups of interferon-γ release assays among health-care workers in contact with patients with TB. Chest. 2012; 142:1461–1468. PMID: 22556318.4. Ringshausen FC, Nienhaus A, Torres Costa J, Knoop H, Schlösser S, Schultze-Werninghaus G, et al. Within-subject variability of Mycobacterium tuberculosis-specific gamma interferon responses in German health care workers. Clin Vaccine Immunol. 2011; 18:1176–1182. PMID: 21593237.5. Metcalfe JZ, Cattamanchi A, McCulloch CE, Lew JD, Ha NP, Graviss EA. Test variability of the QuantiFERON-TB gold in-tube assay in clinical practice. Am J Respir Crit Care Med. 2013; 187:206–211. PMID: 23103734.6. Pai M, Denkinger CM, Kik SV, Rangaka MX, Zwerling A, Oxlade O, et al. Gamma interferon release assays for detection of Mycobacterium tuberculosis infection. Clin Microbiol Rev. 2014; 27:3–20. PMID: 24396134.7. Tagmouti S, Slater M, Benedetti A, Kik SV, Banaei N, Cattamanchi A, et al. Reproducibility of interferon gamma (IFN-γ) release assays. A systematic review. Ann Am Thorac Soc. 2014; 11:1267–1276. PMID: 25188809.8. Woo KS, Choi JL, Kim BR, Kim JE, Kim BG, Lee H, et al. Significance of interferon-gamma response to mitogen in serial QuantiFERON-TB Gold In-Tube assay of routine laboratory practice. Clin Chim Acta. 2014; 430:79–83. PMID: 24389051.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Comparison of Interferon-gamma Assays with the Tuberculin Skin Test in Children
- Performance Evaluation of a New Automated Chemiluminescent Immunoanalyzer-Based Interferon-Gamma Releasing Assay AdvanSure I3 in Comparison With the QuantiFERON-TB Gold In-Tube Assay
- Tuberculin Skin Test and QuantiFERON-TB Gold Assay before and after Treatment for Latent Tuberculosis Infection among Health Care Workers in Local Tertiary Hospital
- The Diagnostic Value of Interferon-gamma Assay in Patients with Active Tuberculosis
- QuantiFERON-TB Gold PLUS versus QuantiFERON- TB Gold In-Tube test for diagnosing tuberculosis infection