Korean J Health Promot.
2016 Dec;16(4):223-230. 10.15384/kjhp.2016.16.4.223.
Analytic Performance Evaluation of Blood Monitoring System G400 according to ISO 15197:2013
- Affiliations
-
- 1Department of Family Medicine, Soonchunhyang University Seoul Hospital, Soonchunhyang University College of Medicine, Seoul, Korea. dryoo@schmc.ac.kr
- KMID: 2367355
- DOI: http://doi.org/10.15384/kjhp.2016.16.4.223
Abstract
- BACKGROUND
Self-monitoring of blood glucose is an important component of therapy for diabetes mellitus. The aim of this study was to evaluate the analytic performance evaluation of blood monitoring system G400 according to ISO 15197:2013.
METHODS
We evaluated the G400 according to the ISO 15197:2013 guideline, we measured precision, accuracy, interference of hematocrit and interfering substances, user performance.
RESULTS
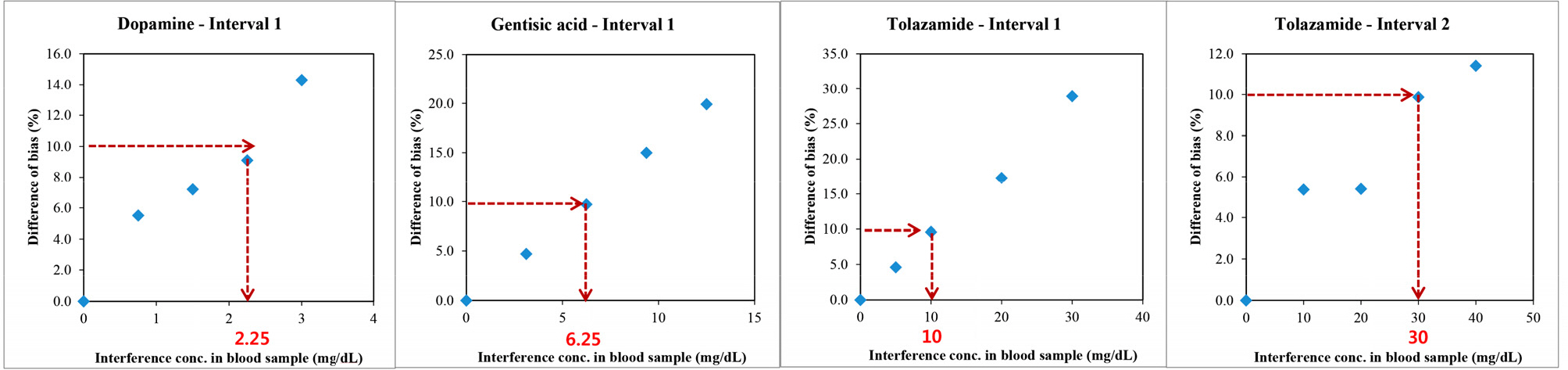
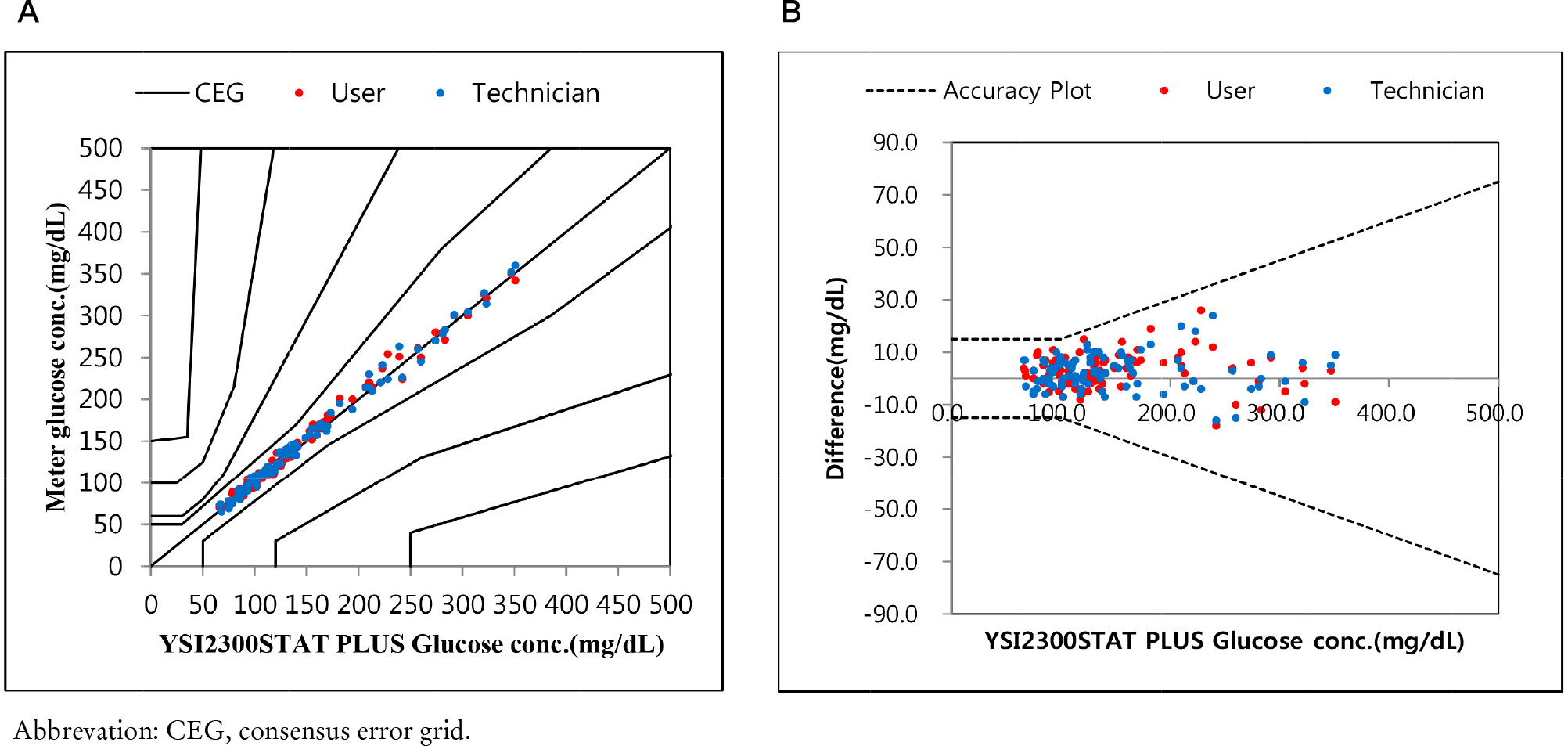
Repeatability and intermediate precision of G400 showed standard deviation 2.7-3.8 mg/dL, 2.4-3.6 mg/dL and coefficient of variation 1.9-2.9% and 1.7-3.7%, respectively. Accuracy measured 98-98.5%, satisfied acceptable criteria. Error grid analysis showed that all results of this study were in zone A. Hematocrit between 20% to 60% did not cause interference. Three of 24 interfering substances were not acceptable criteria, and dose-response evaluation was needed.
CONCLUSIONS
This study showed that G400 was considered reliable results satisfying the ISO 15197:2013 criteria
Figure
Reference
-
References
1. Kim MH, Kim MK, Choi BY, Shin YJ. Educational disparities in the metabolic syndrome in a rapidly changing society–the case of South Korea. Int J Epidemiol. 2005; 34(6):1266–73.
Article2. Huh HJ, Park HD, Lee SY, Kim JW. Evaluation of CareSens(R) N glucometer glucose monitoring system. J Lab Med Qual Assur. 2010; 32(2):229–36.3. Kwon MJ, Lee SY. Evaluation of GLUCOCARD X-METER glucose monitoring system. Korean J Lab Med. 2008; 28(1):8–15.
Article4. Song SH, Park HD, Lee HJ, Chun SH, Park KU, Kim JQ, et al. Evaluation of Gluchec fine glucometer. J Lab Med Qual Assur. 2007; 29(1):187–94.5. Lee SY, Lee NY, Kim JW. Evaluation of 6 glucose testing systems. Korean J Lab Med. 2003; 23(3):170–9.6. Yoo EH, Cho HJ, Ki CS, Lee SY. Evaluation of COSMOsensor glucose monitoring system. Korean J Lab Med. 2006; 26(1):1–8.
Article7. The International Organization for Standardization. In vitro diagnostic test systems – Requirements for blood glucose monitoring systems for self-testing in managing diabetes mellitus. ISO/TC 212/SC. International Standard ISO 15197. Geneva: The International Organization for Standardization;2013.8. Lee JH, Cha YJ. Performance evaluation of BAROZEN H, a networking blood glucose monitoring system for medical institutions. Lab Med Online. 2015; 5(2):69–76.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Performance Evaluation of B. Braun Omnitest 5 Blood Glucose Monitoring System for Self-Monitoring of Blood Glucose
- Performance Evaluation of BAROZEN H, a Networking Blood Glucose Monitoring System for Medical Institutions
- Accuracy of Capillary Blood Glucose Test When Fasting in Diabetes Patients or General Population: Performance Evaluation of G300 Based on ISO 15197:2013 Standards
- Evaluation of the Self-Testing Blood Glucose Monitoring System GlucoDr.S According to ISO 15197:2013 Guidelines
- Performance Evaluation of Glucometer Barozen H Based on ISO 15197 Standards