J Bacteriol Virol.
2016 Dec;46(4):221-230. 10.4167/jbv.2016.46.4.221.
Role of Rotavirus Enterotoxin NSP4 in the Inflammatory Response in Murine Macrophage RAW 264.7 Cells
- Affiliations
-
- 1Department of Microbiology, Chung-Ang University College of Medicine, Seoul, Korea. kimwy@cau.ac.kr
- KMID: 2366867
- DOI: http://doi.org/10.4167/jbv.2016.46.4.221
Abstract
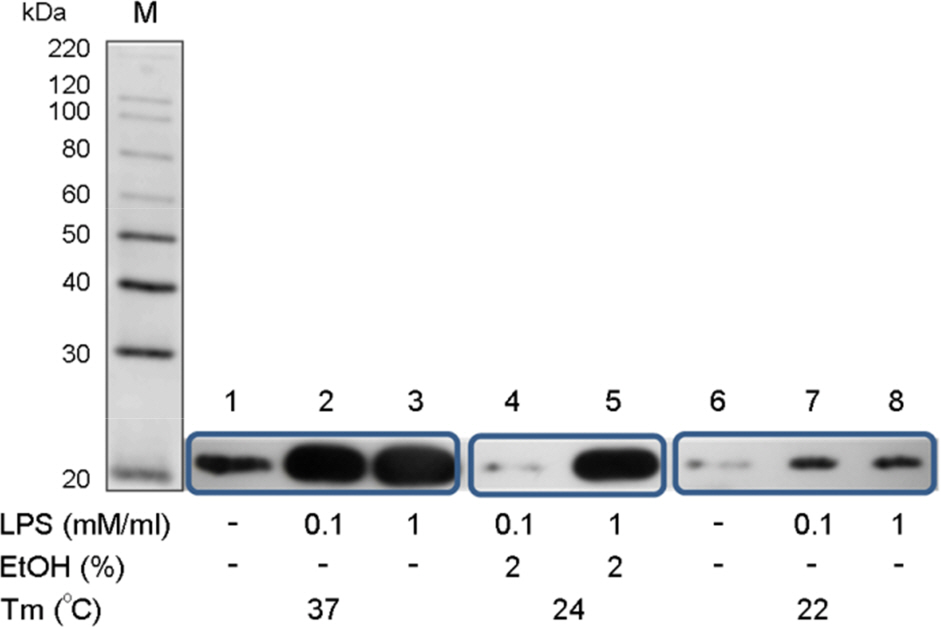
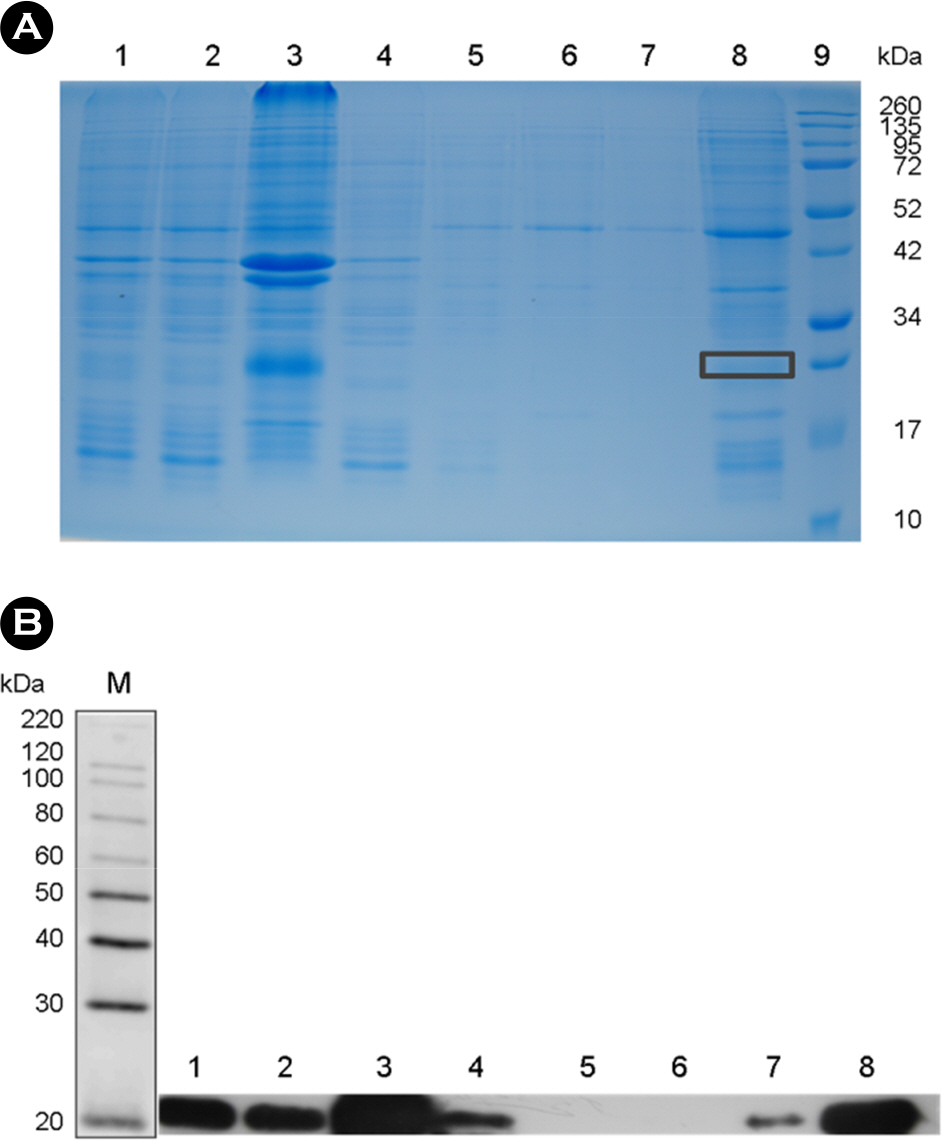
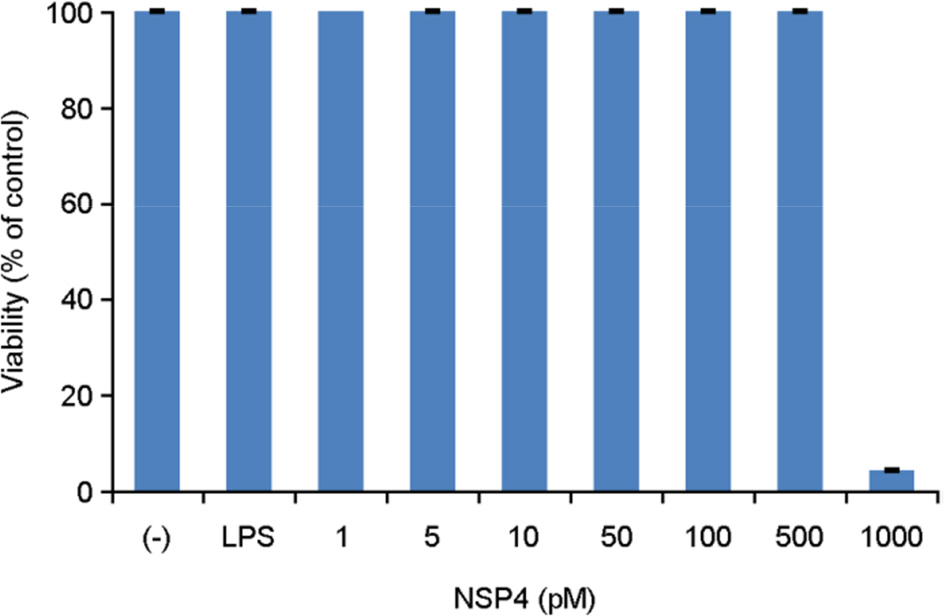
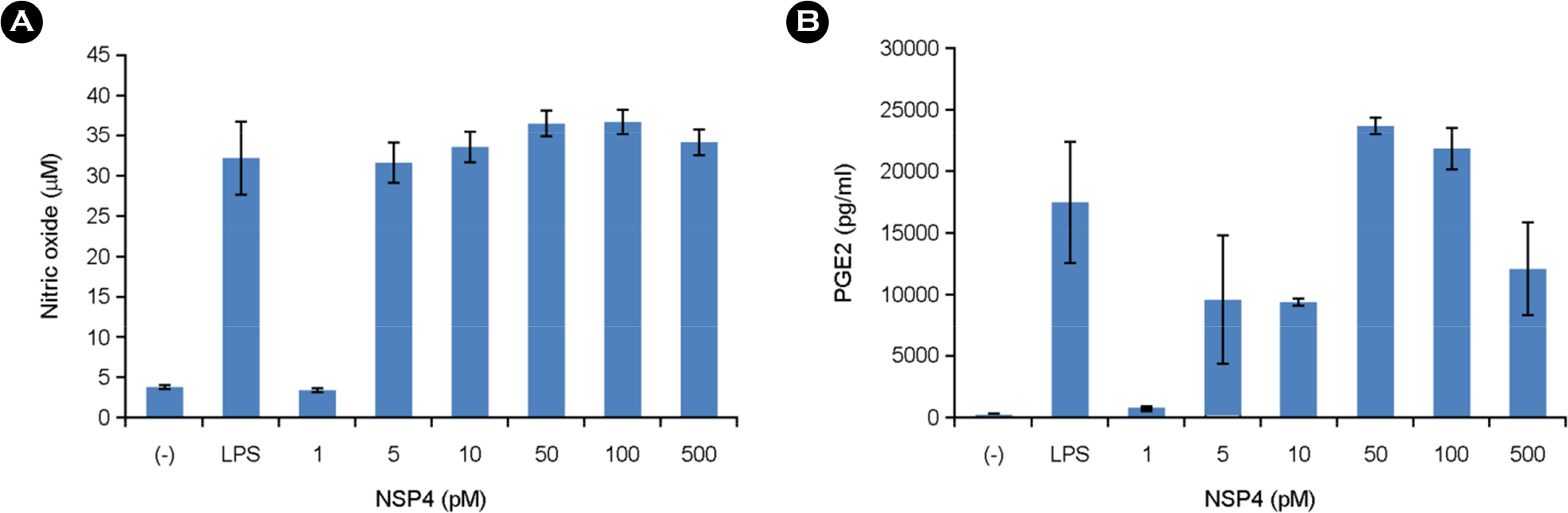
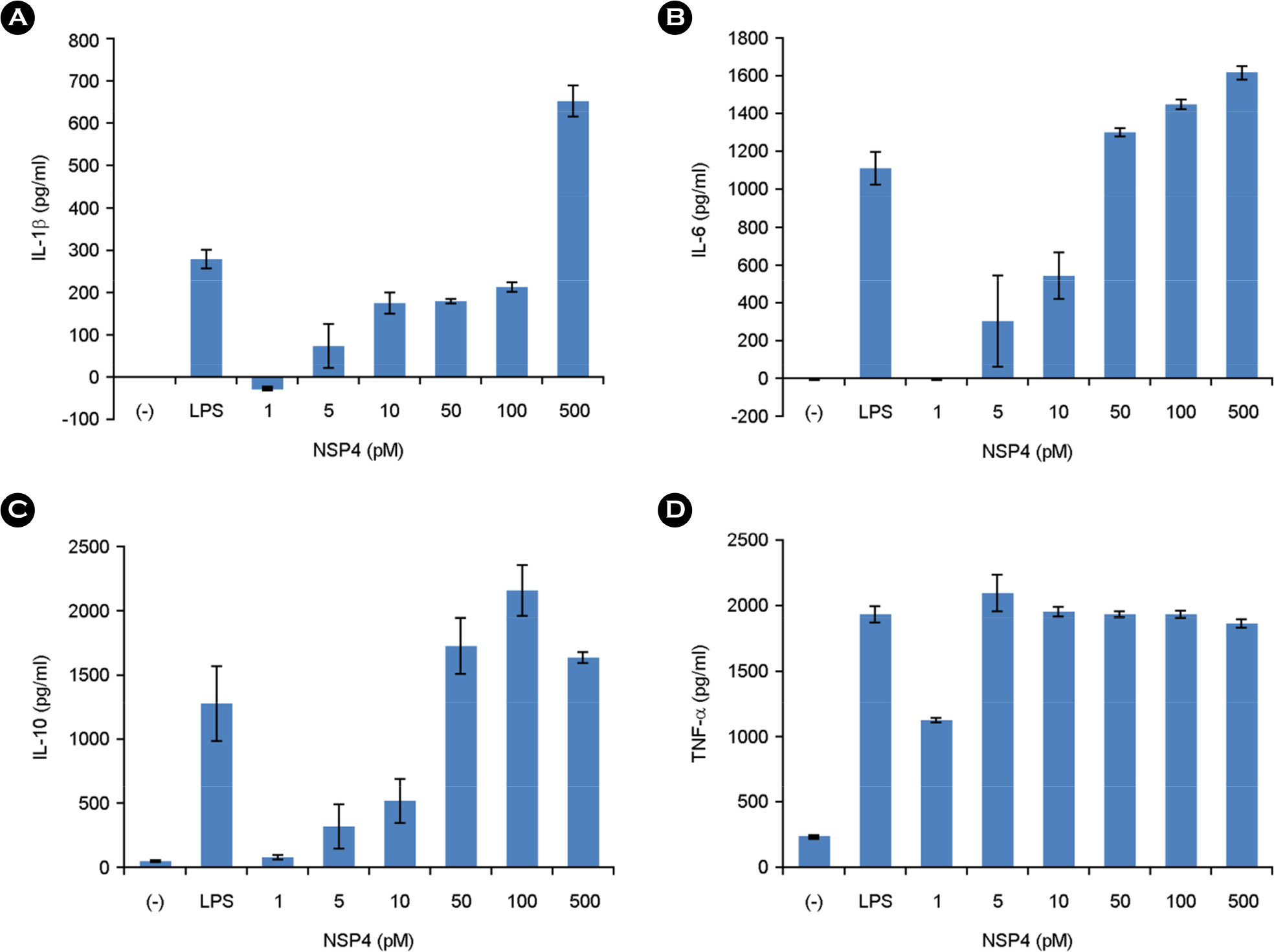
- The rotavirus nonstructural glycoprotein, NSP4, has been identified as the first viral enterotoxin capable of inducing diarrhea. To investigate the biological function of NSP4 in the inflammatory process, a cDNA from human rotavirus (Wa strain) RNA segment 10 was amplified by RT-PCR, cloned into TA vector, and subsequently subcloned into pET23b expression plasmid. The expression of NSP4 protein was determined by SDS-PAGE and Western blotting, then, the protein was purified by affinity chromatography on Ni-NTA-agarose column. The inflammatory effects of NSP4, namely, production of nitric oxide (NO), pro-inflammatory cytokines (IL-1β, IL-6, IL-10, and TNF-α), and prostaglandin E2 (PGE₂), was evaluated using NSP4-stimulated RAW 264.7 murine macrophages and compared with those observed after stimulation with lipopolysaccharide (LPS). The levels of IL-1β, IL-6, and TNF-α were significantly increased, and those of NO and PGE₂ also increased in NSP4-stimulated RAW 264.7 cells. These findings indicate that NSP4 plays an important role in the inflammatory response observed during rotavirus infection.
Keyword
MeSH Terms
-
Blotting, Western
Chromatography, Affinity
Clone Cells
Cytokines
Diarrhea
Dinoprostone
DNA, Complementary
Electrophoresis, Polyacrylamide Gel
Enterotoxins*
Glycoproteins
Humans
Inflammation
Interleukin-10
Interleukin-6
Macrophages*
Nitric Oxide
Plasmids
RAW 264.7 Cells*
RNA
Rotavirus Infections
Rotavirus*
Cytokines
DNA, Complementary
Dinoprostone
Enterotoxins
Glycoproteins
Interleukin-10
Interleukin-6
Nitric Oxide
RNA
Figure
Reference
-
1). Kapikian AZ, Hoshino Y, Chanock RM. Rotaviruses. Knipe D, Howley P, Griffin D, Lamb R, Martin M, Straus S, editors. Fields Virology. 4th ed.Philadelphia: Lippincott, Williams & Wilkins;2001. p. 1787–833.2). Parashar UD, Burton A, Lanata C, Boschi-Pinto C, Shibuya K, Steele D, et al. Global mortality associated with rotavirus disease among children in 2004. J Infect Dis. 2009; 200(Suppl 1):S9–S15.
Article3). Hertel PM, Estes MK. Rotavirus and biliary atresia: can causation be proven? Curr Opin Gastroenterol. 2012; 28:10–7.4). Ball JM, Tian P, Zeng CQ, Morris AP, Estes MK. Age-dependent diarrhea induced by a rotaviral nonstructural glycoprotein. Science. 1996; 272:101–4.
Article5). Dong Y, Zeng CQ, Ball JM, Estes MK, Morris AP. The rotavirus enterotoxin NSP4 mobilizes intracellular calcium in human intestinal cells by stimulating phospholipase C-mediated inositol 1, 4, 5-trisphosphate production. Proc Natl Acad Sci U S A. 1997; 94:3960–5.6). Díaz Y, Peña F, Aristimuño OC, Matteo L, De Agrela M, Chemello ME, et al. Dissecting the Ca(2)(+) entry pathways induced by rotavirus infection and NSP4-EGFP expression in Cos-7 cells. Virus Res. 2012; 167:285–96.
Article7). Rodríguez-Diaz J, López-Andújar P, García-Díaz A, Cuenca J, Montava R, Buesa J. Expression and purification of polyhistidine-tagged rotavirus NSP4 proteins in insect cells. Protein Expr Purif. 2003; 31:207–12.8). Díaz Y, Chemello ME, Peña F, Aristimuño OC, Zambrano JL, Rojas H, et al. Expression of nonstructural rotavirus protein NSP4 mimics Ca2+ homeostasis changes induced by rotavirus infection in cultured cells. J Virol. 2008; 82:11331–43.9). Warfield KL, Blutt SE, Crawford SE, Kang G, Conner ME. Rotavirus infection enhances lipopolysaccharide-induced intussusception in a mouse model. J Virol. 2006; 80:12377–86.
Article10). Ware CF. Network communications: lymphotoxins, LIGHT, and TNF. Annu Rev Immunol. 2005; 23:787–819.
Article11). Ulevitch RJ, Tobias PS. Receptor-dependent mechanisms of cell stimulation by bacterial endotoxin. Annu Rev Immunol. 1995; 13:437–57.
Article12). Harris SG, Padilla J, Koumas L, Ray D, Phipps RP. Prostaglandins as modulators of immunity. Trends Immunol. 2002; 23:144–50.
Article13). Boveris A, Alvarez S, Navarro A. The role of mitochondrial nitric oxide synthase in inflammation and septic shock. Free Radic Biol Med. 2002; 33:1186–93.
Article14). Borghan MA, Mori Y, El-Mahmoudy AB, Ito N, Sugiyama M, Takewaki T, et al. Induction of nitric oxide synthase by rotavirus enterotoxin NSP4: implication for rotavirus pathogenicity. J Gen Virol. 2007; 88:2064–72.
Article15). Ge Y, Mansell A, Ussher JE, Brooks AE, Manning K, Wang CJ, et al. Rotavirus NSP4 Triggers Secretion of Proinflammatory Cytokines from Macrophages via Toll-Like Receptor 2. J Virol. 2013; 87:11160–7.
Article16). Browne EP, Bellamy AR, Taylor JA. Membrane-destabilizing activity of rotavirus NSP4 is mediated by a membrane-proximal amphipathic domain. J Gen Virol. 2000; 81:1955–9.
Article17). Sharifi Z, Yakhchali B, Shahrabadi MS. Expression and one step purification of the full-length biologically active, NSP4 of human rotavirus Wa strain. Int J Mol Med Adv Sci. 2005; 1:206–12.18). Tabandeh F, Sanati MH, Shoja Alsadati SA, Yakhchali B, Khodabandeh M. Evaluation of heat induction strategy for recombinant human growth hormone expression in fed-batch fermentation. Iran J Biotechnology. 2005; 3:24–30.19). Mori Y, Borgan MA, Ito N, Sugiyama M, Minamoto N. Sequential analysis of nonstructural protein NSP4s derived from Group A avian rotaviruses. Virus Res. 2002; 89:145–51.
Article20). Johansen K, Hinkula J, Espinoza F, Levi M, Zeng C, Rudén U, et al. Humoral and cell-mediated immune responses in humans to the NSP4 enterotoxin of rotavirus. J Med Virol. 1999; 59:369–77.
Article21). Iosef C, Chang KO, Azevedo MS, Saif LJ. Systemic and intestinal antibody responses to NSP4 enterotoxin of Wa human rotavirus in a gnotobiotic pig model of human rotavirus disease. J Med Virol. 2002; 68:119–28.
Article22). Rodríguez-Díaz J, Banasaz M, Istrate C, Buesa J, Lundgren O, Espinoza F, et al. Role of nitric oxide during rotavirus infection. J Med Virol. 2006; 78:979–85.
Article23). Allen SR, Jafri M, Donnelly B, McNeal M, Witte D, Bezerra J, et al. Effect of rotavirus strain on the murine model of biliary atresia. J Virol. 2007; 81:1671–9.
Article24). Rauschenfels S, Krassmann M, Al-Masri AN, Verhagen W, Leonhardt J, Kuebler JF, et al. Incidence of hepatotropic viruses in biliary atresia. Eur J Pediatr. 2009; 168:469–76.
Article25). Hertel PM, Crawford SE, Finegold MJ, Estes MK. Osteopontin upregulation in rotavirus-induced murine biliary atresia requires replicating virus but is not necessary for development of biliary atresia. Virology. 2011; 417:281–92.
Article26). Wang W, Donnelly B, Bondoc A, Mohanty SK, McNeal M, Ward R, et al. The rhesus rotavirus gene encoding VP4 is a major determinant in the pathogenesis of biliary atresia in newborn mice. J Virol. 2011; 85:9069–77.
Article27). Feng J, Yang J, Zheng S, Qiu Y, Chai C. Silencing of the rotavirus NSP4 protein decreases the incidence of biliary atresia in murine model. PLoS One. 2011; 6:e23655.
Article28). Yang H, Plösch T, Lisman T, Gouw AS, Porte RJ, Verkade HJ, et al. Inflammation mediated down-regulation of hepatobiliary transporters contributes to intrahepatic cholestasis and liver damage in murine biliary atresia. Pediatr Res. 2009; 66:380–5.
Article29). Vernooy JH, Dentener MA, van Suylen RJ, Buurman WA, Wouters EF. Long-term intratracheal lipopolysaccharide exposure in mice results in chronic lung inflammation and persistent pathology. Am J Respir Cell Mol Biol. 2002; 26:152–9.
Article30). Bondeson J. The mechanisms of action of disease-modifying antirheumatic drugs: a review with emphasis on macrophage signal transduction and the induction of proinflammatory cytokines. Gen Pharmacol. 1997; 29:127–50.
Article31). Kinne RW, Bräuer R, Stuhlmüller B, Palombo-Kinne E, Burmester GR. Macrophages in rheumatoid arthritis. Arthritis Res. 2000; 2:189–202.32). Dayer JM. The process of identifying and understanding cytokines: from basic studies to treating rheumatic diseases. Best Pract Res Clin Rheumatol. 2004; 18:31–45.
Article33). Diehl S, Rincón M. The two faces of IL-6 on Th1/Th2 differentiation. Mol Immunol. 2002; 39:531–6.
Article34). Scheller J, Chalaris A, Schmidt-Arras D, Rose-John S. The proand anti-inflammatory properties of the cytokine interleukin-6. Biochim Biophys Acta. 2011; 1813:878–88.35). Mocellin S, Marincola F, Rossi CR, Nitti D, Lise M. The multifaceted relationship between IL-10 and adaptive immunity: putting together the pieces of a puzzle. Cytokine Growth Factor Rev. 2004; 15:61–76.
Article36). Dace DS, Khan AA, Stark JL, Kelly J, Cross AH, Apte RS. Interleukin-10 overexpression promotes Fas-ligand-dependent chronic macrophage-mediated demyelinating polyneuropathy. PLoS One. 2009; 4:e7121.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Cloning and Sequencing of Nonstructural Protein (NSP4) Gene of Bovine Rotavirus Isolated in Korea
- Anti-inflammatory Effect of Mangosteen (Garcinia mangostana L.) Peel Extract and its Compounds in LPS-induced RAW264.7 Cells
- Genetic Variation in the NSP4 Gene of Human Rotavirus Isolated in Seoul
- Analysis of structure-function relationship in porcine rotavirus A enterotoxin gene
- Lignans and Macrolides from the Leaves of Houttuynia cordata with Inhibitory Activity against NO Production in Murine Macrophage RAW 264.7 Cells