J Korean Med Sci.
2015 Oct;30(10):1453-1458. 10.3346/jkms.2015.30.10.1453.
Effect of Indacaterol on Cough and Phlegm in Chronic Obstructive Pulmonary Disease Patients: A Meta-Analysis of Five Randomized Controlled Trials
- Affiliations
-
- 1Department of Critical Care, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 2Department of Pulmonary and Critical Care Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. ymoh55@amc.seoul.kr
- 3Clinical Research Center for Chronic Obstructive Airway Diseases, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- 4Department of Internal Medicine, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- KMID: 2344178
- DOI: http://doi.org/10.3346/jkms.2015.30.10.1453
Abstract
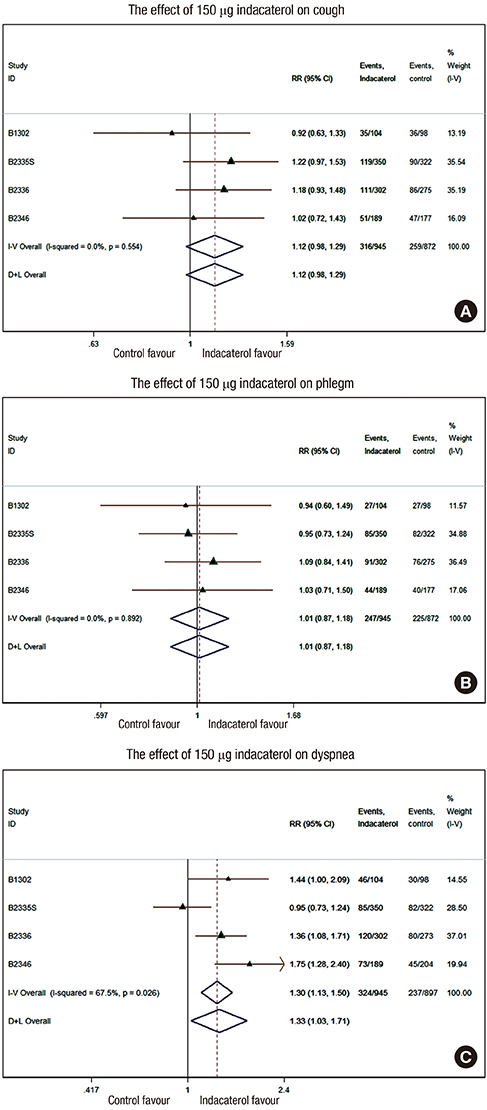
- We investigated the effects of indacaterol on cough and phlegm in patients with stable chronic obstructive pulmonary disease (COPD). We performed a meta-analysis with five randomized controlled trials (RCTs) of indacaterol in stable COPD patients. The symptom severity was defined using the St. George's Respiratory Questionnaire (SGRQ). We analyzed patients treated with 150 microg (n = 945) and 300 microg (n = 832) out of 3,325 patients who completed the SGRQ from five RCTs. After a 12-week treatment of 150 microg indacaterol, cough improvement was reported in 36.5% (316/866) of patients treated with indacaterol vs. 32.2% (259/804) patients treated with placebo (Relative Ratio [RR], 1.13; 95% confidence interval [CI], 0.99-1.29). Phlegm improvement was reported in 31.0% (247/798) of patients treated with indacaterol vs. 30.6% (225/736) of patients treated with placebo (RR, 1.01; 95% CI, 0.87-1.18). Dyspnea improvement was reported in 39.5% (324/820) of patients treated with indacaterol vs. 31.5% (237/753) patients treated with placebo (RR, 1.33; 95% CI, 1.03-1.71; P = 0.001, I2 = 55.1%). Only dyspnea improvement was significant compared to placebo even at the 300 microg indacaterol dose. Compared to placebo, a 12-week treatment of the long-acting beta-agonist, indacaterol might not have a significant effect on cough or phlegm in stable COPD.
MeSH Terms
-
Administration, Inhalation
Anti-Bacterial Agents/therapeutic use
Bronchodilator Agents/*therapeutic use
Cough/*drug therapy
Dyspnea/*drug therapy
Forced Expiratory Volume/drug effects
Humans
Indans/*therapeutic use
Placebos/administration & dosage
Pulmonary Disease, Chronic Obstructive/*drug therapy
Quinolones/*therapeutic use
Sputum/*drug effects
Surveys and Questionnaires
Treatment Outcome
Anti-Bacterial Agents
Bronchodilator Agents
Indans
Placebos
Quinolones
Figure
Reference
-
1. Fletcher C, Peto R. The natural history of chronic airflow obstruction. Br Med J. 1977; 1:1645–1648.2. Lee JS, Rhee CK, Yoo KH, Lee JH, Yoon HI, Kim TH, Kim WJ, Lee J, Lim SY, Park TS, et al. Three-month treatment response and exacerbation in chronic obstructive pulmonary disease. J Korean Med Sci. 2015; 30:54–59.3. Figarska SM, Boezen HM, Vonk JM. Dyspnea severity, changes in dyspnea status and mortality in the general population: the Vlagtwedde/Vlaardingen study. Eur J Epidemiol. 2012; 27:867–876.4. Nishimura K, Izumi T, Tsukino M, Oga T. Dyspnea is a better predictor of 5-year survival than airway obstruction in patients with COPD. Chest. 2002; 121:1434–1440.5. Müllerová H, Lu C, Li H, Tabberer M. Prevalence and burden of breathlessness in patients with chronic obstructive pulmonary disease managed in primary care. PLoS One. 2014; 9:e85540.6. Rennard S, Decramer M, Calverley PM, Pride NB, Soriano JB, Vermeire PA, Vestbo J. Impact of COPD in North America and Europe in 2000: subjects' perspective of Confronting COPD International Survey. Eur Respir J. 2002; 20:799–805.7. Calverley PM. Cough in chronic obstructive pulmonary disease: is it important and what are the effects of treatment? Cough. 2013; 9:17.8. Burgel PR, Nesme-Meyer P, Chanez P, Caillaud D, Carré P, Perez T, Roche N. Initiatives Bronchopneumopathie Chronique Obstructive Scientific Committee. Cough and sputum production are associated with frequent exacerbations and hospitalizations in COPD subjects. Chest. 2009; 135:975–982.9. Beeh KM, Wagner F, Khindri S, Drollmann AF. Effect of indacaterol on dynamic lung hyperinflation and breathlessness in hyperinflated patients with COPD. COPD. 2011; 8:340–345.10. Watz H, Krippner F, Kirsten A, Magnussen H, Vogelmeier C. Indacaterol improves lung hyperinflation and physical activity in patients with moderate chronic obstructive pulmonary disease--a randomized, multicenter, double-blind, placebo-controlled study. BMC Pulm Med. 2014; 14:158.11. Han J, Dai L, Zhong N. Indacaterol on dyspnea in chronic obstructive pulmonary disease: a systematic review and meta-analysis of randomized placebo-controlled trials. BMC Pulm Med. 2013; 13:26.12. To Y, Kinoshita M, Lee SH, Hang LW, Ichinose M, Fukuchi Y, Kitawaki T, Okino N, Prasad N, Lawrence D, et al. Assessing efficacy of indacaterol in moderate and severe COPD patients: a 12-week study in an Asian population. Respir Med. 2012; 106:1715–1721.13. Dahl R, Chung KF, Buhl R, Magnussen H, Nonikov V, Jack D, Bleasdale P, Owen R, Higgins M, Kramer B, et al. INVOLVE (INdacaterol: Value in COPD: Longer Term Validation of Efficacy and Safety) Study Investigators. Efficacy of a new once-daily long-acting inhaled beta2-agonist indacaterol versus twice-daily formoterol in COPD. Thorax. 2010; 65:473–479.14. Donohue JF, Fogarty C, Lötvall J, Mahler DA, Worth H, Yorgancioglu A, Iqbal A, Swales J, Owen R, Higgins M, et al. INHANCE Study Investigators. Once-daily bronchodilators for chronic obstructive pulmonary disease: indacaterol versus tiotropium. Am J Respir Crit Care Med. 2010; 182:155–162.15. Kornmann O, Dahl R, Centanni S, Dogra A, Owen R, Lassen C, Kramer B. INLIGHT-2 (Indacaterol Efficacy Evaluation Using 150-µg Doses with COPD Patients) study investigators. Indacaterol Efficacy Evaluation Using 150-μg Doses with COPD Patients) study investigators. Once-daily indacaterol versus twice-daily salmeterol for COPD: a placebo-controlled comparison. Eur Respir J. 2011; 37:273–279.16. Feldman G, Siler T, Prasad N, Jack D, Piggott S, Owen R, Higgins M, Kramer B. INLIGHT 1 study group. Efficacy and safety of indacaterol 150 microg once-daily in COPD: a double-blind, randomised, 12-week study. BMC Pulm Med. 2010; 10:11.17. DerSimonian R, Kacker R. Random-effects model for meta-analysis of clinical trials: an update. Contemp Clin Trials. 2007; 28:105–114.18. Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002; 21:1539–1558.19. Leidy NK, Murray LT. Patient-reported outcome (PRO) measures for clinical trials of COPD: the EXACT and E-RS. COPD. 2013; 10:393–398.20. Chung VC, Ma PH, Hui DS, Tam WW, Tang JL. Indacaterol for chronic obstructive pulmonary disease: systematic review and meta-analysis. PLoS One. 2013; 8:e70784.21. Jones PW, Quirk FH, Baveystock CM, Littlejohns P. A self-complete measure of health status for chronic airflow limitation. The St Georges Respiratory Questionnaire. Am Rev Respir Dis. 1992; 145:1321–1327.22. Chang JA, Curtis JR, Patrick DL, Raghu G. Assessment of health-related quality of life in patients with interstitial lung disease. Chest. 1999; 116:1175–1182.23. Devalia JL, Sapsford RJ, Rusznak C, Toumbis MJ, Davies RJ. The effects of salmeterol and salbutamol on ciliary beat frequency of cultured human bronchial epithelial cells, in vitro. Pulm Pharmacol. 1992; 5:257–263.24. Devalia JL, Sapsford RJ, Rusznak C, Toumbis MJ, Davies RJ. The effects of salmeterol and salbutamol on ciliary beat frequency of cultured human bronchial epithelial cells, in vitro. Pulm Pharmacol. 1992; 5:257–263.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Factors Associated with Indacaterol Response in Tuberculosis-Destroyed Lung with Airflow Limitation
- Meta-analysis of the Effect of a Pulmonary Rehabilitation Program on Respiratory Muscle Strength in Patients with Chronic Obstructive Pulmonary Disease
- Systematic Review and Meta-Analysis of Pulmonary Hypertension Specific Therapy for Exercise Capacity in Chronic Obstructive Pulmonary Disease
- Safety and Effectiveness of Indacaterol in Chronic Obstructive Pulmonary Disease Patients in South Korea
- Efficacy and Safety of Gabapentin in the Treatment of Chronic Cough: A Systematic Review