Korean J Urol.
2011 Nov;52(11):776-781.
Evaluation of the Optimal Frequency of and Pretreatment with Shock Waves in Patients with Renal Stones
- Affiliations
-
- 1Department of Urology, Chung-Ang University College of Medicine, Seoul, Korea. Moon2580@cau.ac.kr
Abstract
- PURPOSE
Many studies have been carried out to increase the success rate of shock wave lithotripsy (SWL) and to reduce renal injury. We investigated the success rate after one session as well as urine N-acetyl-beta-d-glucosaminidase (NAG) levels for the evaluation of renal injury according to shock wave frequency and pretreatment with low-energy shock waves during SWL.
MATERIALS AND METHODS
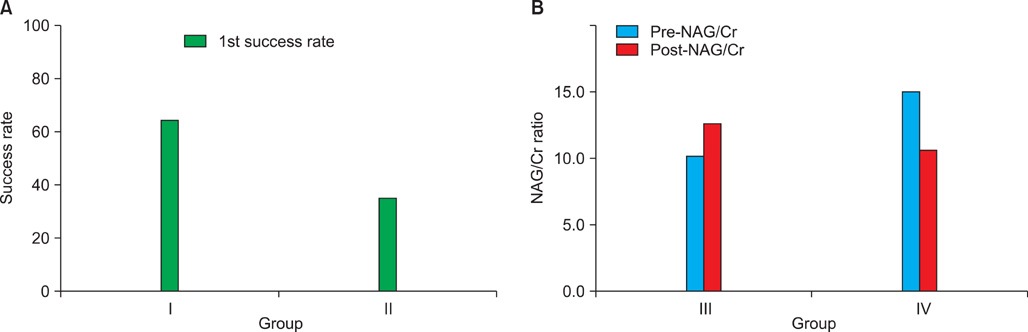
The study targeted 48 patients with renal stones who had undergone SWL. Patients were sequentially allocated into four groups according to shock wave frequency (60 or 120 shocks/min) and whether pretreatment had occurred. We documented total SWL operating number, success rate after first SWL, urine NAG, compliance, and the total cost for each patient.
RESULTS
There were 32 males and 16 females with an average age of 51.6 years. The average stone size was 7.06 mm, and there was no significant difference in stone size between the groups. The data showed that patients treated with a frequency of 60 shocks/min had a lower mean number of SWL sessions, 1.36 sessions for 60 shocks/min and 2.0 sessions for 120 shocks/min, respectively, which was statistically significant (p<0.05). When comparing NAG/creatinin ratios before and after SWL between those with and without pretreatment, there was no significant difference according to pretreatment (p=0.406).
CONCLUSIONS
SWL treatment at a frequency of 60 shocks/min yielded better outcomes, such as a lower number of SWL sessions, and had an increased success rate compared with SWL at 120 shocks/min. On the other hand, pretreatment did not impact renal injury. Therefore, SWL treatment at a frequency of 60 shocks/min could improve treatment efficacy more than that for SWL at 120 shocks/min.
Keyword
MeSH Terms
Figure
Reference
-
1. Skolarikos A, Alivizatos G, de la Rosette J. Extracorporeal shock wave lithotripsy 25 years later: complications and their prevention. Eur Urol. 2006. 50:981–990.2. Albala DM, Assimos DG, Clayman RV, Denstedt JD, Grasso M, Gutierrez-Aceves J, et al. Lower pole I: a prospective randomized trial of extracorporeal shock wave lithotripsy and percutaneous nephrostolithotomy for lower pole nephrolithiasis-initial results. J Urol. 2001. 166:2072–2080.3. Delius M, Enders G, Xuan ZR, Liebich HG, Brendel W. Biological effects of shock waves: kidney damage by shock waves in dogs--dose dependence. Ultrasound Med Biol. 1988. 14:117–122.4. Zhou Y, Cocks FH, Preminger GM, Zhong P. The effect of treatment strategy on stone comminution efficiency in shock wave lithotripsy. J Urol. 2004. 172:349–354.5. Pace KT, Ghiculete D, Harju M, Honey RJ. University of Toronto Lithotripsy Associates. Shock wave lithotripsy at 60 or 120 shocks per minute: a randomized, double-blind trial. J Urol. 2005. 174:595–599.6. Chacko J, Moore M, Sankey N, Chandhoke PS. Does a slower treatment rate impact the efficacy of extracorporeal shock wave lithotripsy for solitary kidney or ureteral stones? J Urol. 2006. 175:1370–1373.7. Greenstein A, Matzkin H. Does the rate of extracorporeal shock wave delivery affect stone fragmentation? Urology. 1999. 54:430–432.8. Yilmaz E, Batislam E, Basar M, Tuglu D, Mert C, Basar H. Optimal frequency in extracorporeal shock wave lithotripsy: prospective randomized study. Urology. 2005. 66:1160–1164.9. Willis LR, Evan AP, Connors BA, Handa RK, Blomgren PM, Lingeman JE. Prevention of lithotripsy-induced renal injury by pretreating kidneys with low-energy shock waves. J Am Soc Nephrol. 2006. 17:663–673.10. Csáthy L, Pócsi I. Urinary N-acetyl-beta-D-glucosaminidase determination in newborns and children: methods and diagnostic applications. Eur J Clin Chem Clin Biochem. 1995. 33:575–587.11. Skalova S, Chladek J. Urinary N-acetyl-beta-D-glucosaminidase activity in healthy children. Nephrology (Carlton). 2004. 9:19–21.12. Turecky L, Uhlikova E. Diagnostic significance of urinary enzymes in nephrology. Bratisl Lek Listy. 2003. 104:27–31.13. Kim SW, Sul CK. Evaluation of renal injury in patients with renal stone after ESWL. Korean J Urol. 1996. 37:325–330.14. Koo V, Beattie I, Young M. Improved cost-effectiveness and efficiency with a slower shockwave delivery rate. BJU Int. 2010. 105:692–696.15. Kim JH, Moon YT. Predicting the therapeutic effect of extracorporeal shockwave lithotripsy by non-enhanced computed tomography in renal stones. Korean J Urol. 2008. 49:252–256.16. Semins MJ, Trock BJ, Matlaga BR. The effect of shock wave rate on the outcome of shock wave lithotripsy: a meta-analysis. J Urol. 2008. 179:194–197.17. Talic RF, Rabah DM. Effect of modification of shock-wave delivery on stone fragmentation. Curr Opin Urol. 2006. 16:83–87.18. Zeman RK, Davros WJ, Garra BS, Horii SC. Cavitation effects during lithotripsy. Part I. Results of in vitro experiments. Radiology. 1990. 177:157–161.19. Choi MJ, Coleman AJ, Saunders JE. The influence of fluid properties and pulse amplitude on bubble dynamics in the field of a shock wave lithotripter. Phys Med Biol. 1993. 38:1561–1573.20. Karlsen SJ, Smevik B, Hovig T. Acute morphological changes in canine kidneys after exposure to extracorporeal shock waves. A light and electron microscopic study. Urol Res. 1991. 19:105–115.21. Recker F, Hofmann W, Bex A, Tscholl R. Quantitative determination of urinary marker proteins: a model to detect intrarenal bioeffects after extracorporeal lithotripsy. J Urol. 1992. 148:1000–1006.22. Delvecchio F, Auge BK, Munver R, Brown SA, Brizuela R, Zhong P, et al. Shock wave lithotripsy causes ipsilateral renal injury remote from the focal point: the role of regional vasoconstriction. J Urol. 2003. 169:1526–1529.23. Connors BA, Evan AP, Willis LR, Blomgren PM, Lingeman JE, Fineberg NS. The effect of discharge voltage on renal injury and impairment caused by lithotripsy in the pig. J Am Soc Nephrol. 2000. 11:310–318.24. Karlin GS, Schulsinger D, Urivetsky M, Smith AD. Absence of persisting parenchymal damage after extracorporeal shock wave lithotripsy as judged by excretion of renal tubular enzymes. J Urol. 1990. 144:13–14.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Piezoelectrically Generated Extracorporeal Shock Wave Lithotripsy for Fragmentation of Extrahepatic Bile Duct Stones
- Evaluation of urinary enzyme levels after piezoelectric extracorporeal shock wave lithotripsy for renal stones
- Extracorporeal shock wave lithotripsy of intrahepatic stones with piezoelectric lithotriptor: in vitro study
- The Efficacy of Extracorporeal Shock Wave Lithotripsy(ESWL) for Mid-ureter Stone
- Evaluation of Renal Injury in Patients with Renal Stone after ESWL