Endocrinol Metab.
2015 Dec;30(4):429-435. 10.3803/EnM.2015.30.4.429.
Emerging Therapies for Osteoporosis
- Affiliations
-
- 1Oregon Osteoporosis Center, Portland, OR, USA. mmcclung@orost.com
- KMID: 2169649
- DOI: http://doi.org/10.3803/EnM.2015.30.4.429
Abstract
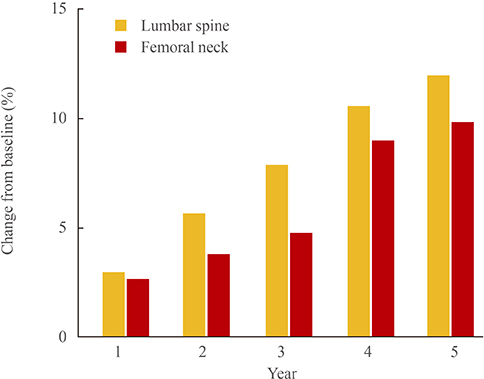
- Although several effective therapies are available for the treatment of osteoporosis in postmenopausal women and older men, there remains a need for the development of even more effective and acceptable drugs. Several new drugs that are in late-stage clinical development will be discussed. Abaloparatide (recombinant parathyroid hormone related peptide [PTHrP] analogue) has anabolic activity like teriparatide. Recent data from the phase 3 fracture prevention trial demonstrate that this agent is effective in reducing fracture risk. Inhibiting cathepsin K reduces bone resorption without decreasing the numbers or activity of osteoclasts, thereby preserving or promoting osteoblast function. Progressive increases in bone mineral density (BMD) have been observed over 5 years. Early data suggest that odanacatib effectively reduces fracture risk. Lastly, inhibiting sclerostin with humanized antibodies promotes rapid, substantial but transient increases in bone formation while inhibiting bone resorption. Marked increases in BMD have been observed in phase 2 studies. Fracture prevention studies are underway. The new therapies with novel and unique mechanisms of action may, alone or in combination, provide more effective treatment options for our patients.
MeSH Terms
Figure
Cited by 1 articles
-
Potential Biomarkers to Improve the Prediction of Osteoporotic Fractures
Beom-Jun Kim, Seung Hun Lee, Jung-Min Koh
Endocrinol Metab. 2020;35(1):55-63. doi: 10.3803/EnM.2020.35.1.55.
Reference
-
1. Black DM, Delmas PD, Eastell R, Reid IR, Boonen S, Cauley JA, et al. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med. 2007; 356:1809–1822.2. Cummings SR, San Martin J, McClung MR, Siris ES, Eastell R, Reid IR, et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009; 361:756–765.3. Hodsman AB, Bauer DC, Dempster DW, Dian L, Hanley DA, Harris ST, et al. Parathyroid hormone and teriparatide for the treatment of osteoporosis: a review of the evidence and suggested guidelines for its use. Endocr Rev. 2005; 26:688–703.4. Neer RM, Arnaud CD, Zanchetta JR, Prince R, Gaich GA, Reginster JY, et al. Effect of parathyroid hormone (1-34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med. 2001; 344:1434–1441.5. Greenspan SL, Bone HG, Ettinger MP, Hanley DA, Lindsay R, Zanchetta JR, et al. Effect of recombinant human parathyroid hormone (1-84) on vertebral fracture and bone mineral density in postmenopausal women with osteoporosis: a randomized trial. Ann Intern Med. 2007; 146:326–339.6. Lagari VS, McAninch E, Baim S. Considerations regarding adherence of anti-osteoporosis therapy. Postgrad Med. 2015; 127:92–98.7. McClung M, Harris ST, Miller PD, Bauer DC, Davison KS, Dian L, et al. Bisphosphonate therapy for osteoporosis: benefits, risks, and drug holiday. Am J Med. 2013; 126:13–20.8. Dean T, Vilardaga JP, Potts JT Jr, Gardella TJ. Altered selectivity of parathyroid hormone (PTH) and PTH-related protein (PTHrP) for distinct conformations of the PTH/PTHrP receptor. Mol Endocrinol. 2008; 22:156–166.9. Cupp ME, Nayak SK, Adem AS, Thomsen WJ. Parathyroid hormone (PTH) and PTH-related peptide domains contributing to activation of different PTH receptor-mediated signaling pathways. J Pharmacol Exp Ther. 2013; 345:404–418.10. Rodriguez-de la Rosa L, Lopez-Herradon A, Portal-Nunez S, Murillo-Cuesta S, Lozano D, Cediel R, et al. Treatment with N- and C-terminal peptides of parathyroid hormone-related protein partly compensate the skeletal abnormalities in IGF-I deficient mice. PLoS One. 2014; 9:e87536.11. Horwitz MJ, Augustine M, Khan L, Martin E, Oakley CC, Carneiro RM, et al. A comparison of parathyroid hormone-related protein (1-36) and parathyroid hormone (1-34) on markers of bone turnover and bone density in postmenopausal women: the PrOP study. J Bone Miner Res. 2013; 28:2266–2276.12. Leder BZ, O'Dea LS, Zanchetta JR, Kumar P, Banks K, McKay K, et al. Effects of abaloparatide, a human parathyroid hormone-related peptide analog, on bone mineral density in postmenopausal women with osteoporosis. J Clin Endocrinol Metab. 2015; 100:697–706.13. Miller PD, Leder BZ, Hattersley G, Lau E, Alexandersen P, Hala T, et al. LB-OR01-3 Effects of abaloparatide on vertebral and non-vertebral fracture incidence in postmenopausal women with osteoporosis: results of the phase 3 active trial. In : The Endocrine Society's 97th Annual Meeting & Expo; 2015 Mar 5-8; San Diego, CA. 2015.14. Rodan SB, Duong LT. Cathepsin K: a new molecular target for osteoporosis. IBMS BoneKEy. 2008; 5:16–24.15. Costa AG, Cusano NE, Silva BC, Cremers S, Bilezikian JP. Cathepsin K: its skeletal actions and role as a therapeutic target in osteoporosis. Nat Rev Rheumatol. 2011; 7:447–456.16. Pennypacker B, Shea M, Liu Q, Masarachia P, Saftig P, Rodan S, et al. Bone density, strength, and formation in adult cathepsin K (-/-) mice. Bone. 2009; 44:199–207.17. Gauthier JY, Chauret N, Cromlish W, Desmarais S, Duong LT, Falgueyret JP, et al. The discovery of odanacatib (MK-0822), a selective inhibitor of cathepsin K. Bioorg Med Chem Lett. 2008; 18:923–928.18. Stroup GB, Kumar S, Jerome CP. Treatment with a potent cathepsin K inhibitor preserves cortical and trabecular bone mass in ovariectomized monkeys. Calcif Tissue Int. 2009; 85:344–355.19. Jerome C, Missbach M, Gamse R. Balicatib, a cathepsin K inhibitor, stimulates periosteal bone formation in monkeys. Osteoporos Int. 2011; 22:3001–3011.20. Masarachia PJ, Pennypacker BL, Pickarski M, Scott KR, Wesolowski GA, Smith SY, et al. Odanacatib reduces bone turnover and increases bone mass in the lumbar spine of skeletally mature ovariectomized rhesus monkeys. J Bone Miner Res. 2012; 27:509–523.21. Pennypacker BL, Duong LT, Cusick TE, Masarachia PJ, Gentile MA, Gauthier JY, et al. Cathepsin K inhibitors prevent bone loss in estrogen-deficient rabbits. J Bone Miner Res. 2011; 26:252–262.22. Cusick T, Chen CM, Pennypacker BL, Pickarski M, Kimmel DB, Scott BB, et al. Odanacatib treatment increases hip bone mass and cortical thickness by preserving endocortical bone formation and stimulating periosteal bone formation in the ovariectomized adult rhesus monkey. J Bone Miner Res. 2012; 27:524–537.23. Pennypacker BL, Chen CM, Zheng H, Shih MS, Belfast M, Samadfam R, et al. Inhibition of cathepsin K increases modeling-based bone formation, and improves cortical dimension and strength in adult ovariectomized monkeys. J Bone Miner Res. 2014; 29:1847–1858.24. Lotinun S, Kiviranta R, Matsubara T, Alzate JA, Neff L, Luth A, et al. Osteoclast-specific cathepsin K deletion stimulates S1P-dependent bone formation. J Clin Invest. 2013; 123:666–681.25. Runger TM, Adami S, Benhamou CL, Czerwinski E, Farrerons J, Kendler DL, et al. Morphea-like skin reactions in patients treated with the cathepsin K inhibitor balicatib. J Am Acad Dermatol. 2012; 66:e89–e96.26. Falgueyret JP, Desmarais S, Oballa R, Black WC, Cromlish W, Khougaz K, et al. Lysosomotropism of basic cathepsin K inhibitors contributes to increased cellular potencies against off-target cathepsins and reduced functional selectivity. J Med Chem. 2005; 48:7535–7543.27. Eastell R, Nagase S, Ohyama M, Small M, Sawyer J, Boonen S, et al. Safety and efficacy of the cathepsin K inhibitor ONO-5334 in postmenopausal osteoporosis: the OCEAN study. J Bone Miner Res. 2011; 26:1303–1312.28. Bone HG, McClung MR, Roux C, Recker RR, Eisman JA, Verbruggen N, et al. Odanacatib, a cathepsin-K inhibitor for osteoporosis: a two-year study in postmenopausal women with low bone density. J Bone Miner Res. 2010; 25:937–947.29. Langdahl B, Binkley N, Bone H, Gilchrist N, Resch H, Rodriguez Portales J, et al. Odanacatib in the treatment of postmenopausal women with low bone mineral density: five years of continued therapy in a phase 2 study. J Bone Miner Res. 2012; 27:2251–2258.30. Eisman JA, Bone HG, Hosking DJ, McClung MR, Reid IR, Rizzoli R, et al. Odanacatib in the treatment of postmenopausal women with low bone mineral density: three-year continued therapy and resolution of effect. J Bone Miner Res. 2011; 26:242–251.31. Bone HG, Dempster DW, Eisman JA, Greenspan SL, McClung MR, Nakamura T, et al. Odanacatib for the treatment of postmenopausal osteoporosis: development history and design and participant characteristics of LOFT, the Long-Term Odanacatib Fracture Trial. Osteoporos Int. 2015; 26:699–712.32. McClung MR, Langdahl B, Papapoulos S, Saag KG, Adami S, Bone HG, et al. Odanacatib anti-fracture efficacy and safety in postmenopausal women with osteoporosis: results from the Phase III Long-term Odanacatib Fracture Trial (LOFT). Arthritis Rheumatol. 2014; 66:Suppl 10. S987.33. van Lierop AH, Hamdy NA, van Bezooijen RL, Lowik CW, Papapoulos SE. The role of sclerostin in the pathophysiology of sclerosing bone dysplasias. Clin Rev Bone Miner Metab. 2012; 10:108–116.34. Gardner JC, van Bezooijen RL, Mervis B, Hamdy NA, Lowik CW, Hamersma H, et al. Bone mineral density in sclerosteosis; affected individuals and gene carriers. J Clin Endocrinol Metab. 2005; 90:6392–6395.35. Li X, Ominsky MS, Warmington KS, Morony S, Gong J, Cao J, et al. Sclerostin antibody treatment increases bone formation, bone mass, and bone strength in a rat model of postmenopausal osteoporosis. J Bone Miner Res. 2009; 24:578–588.36. Li X, Ominsky MS, Niu QT, Sun N, Daugherty B, D'Agostin D, et al. Targeted deletion of the sclerostin gene in mice results in increased bone formation and bone strength. J Bone Miner Res. 2008; 23:860–869.37. Ominsky MS, Vlasseros F, Jolette J, Smith SY, Stouch B, Doellgast G, et al. Two doses of sclerostin antibody in cynomolgus monkeys increases bone formation, bone mineral density, and bone strength. J Bone Miner Res. 2010; 25:948–959.38. McClung MR, Grauer A, Boonen S, Bolognese MA, Brown JP, Diez-Perez A, et al. Romosozumab in postmenopausal women with low bone mineral density. N Engl J Med. 2014; 370:412–420.39. Recker RR, Benson CT, Matsumoto T, Bolognese MA, Robins DA, Alam J, et al. A randomized, double-blind phase 2 clinical trial of blosozumab, a sclerostin antibody, in postmenopausal women with low bone mineral density. J Bone Miner Res. 2015; 30:216–224.