Ann Surg Treat Res.
2016 Jan;90(1):49-52. 10.4174/astr.2016.90.1.49.
Currarino triad with Mullerian duct anomaly in mother and daughter without MNX1 gene mutation
- Affiliations
-
- 1Department of Pediatric Surgery, Pusan National University Yangsan Hospital, Yangsan, Korea.
- 2Department of Pediatric Surgery, Seoul National University Children's Hospital, Seoul, Korea. spkhy02@snu.ac.kr
- KMID: 2149932
- DOI: http://doi.org/10.4174/astr.2016.90.1.49
Abstract
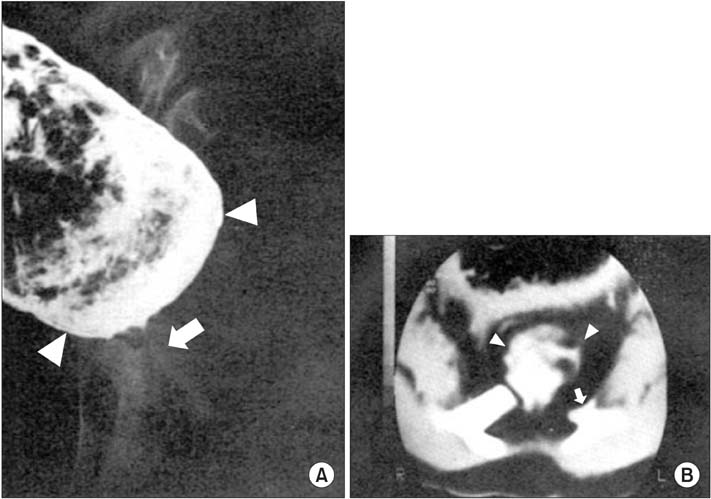
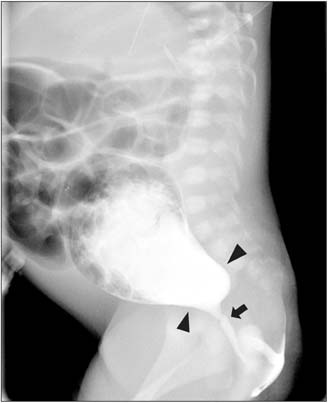
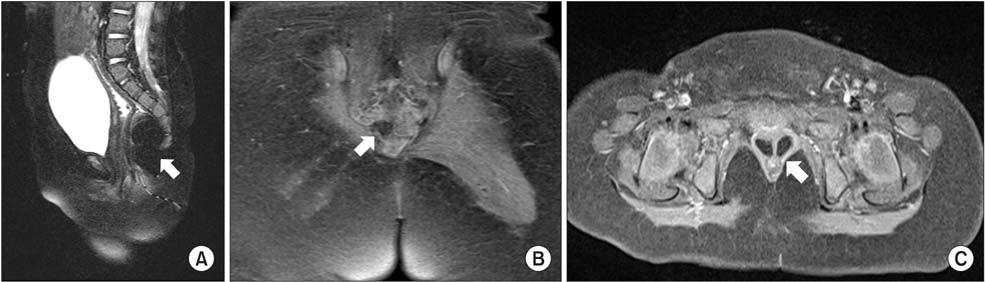
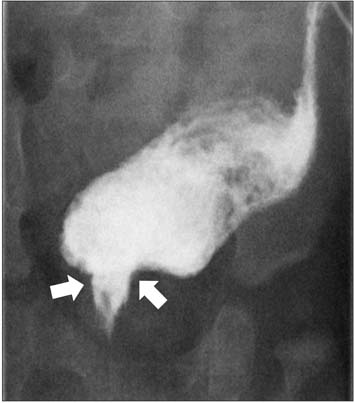
- The Currarino triad is a unique complex of congenital caudal anomalies, including anorectal malformation, sacral bony defect and presacral mass. This triad may be associated with Mullerian duct anomalies, such as duplication of the vagina and uterus. Each of these diseases has a familial tendency and sometimes coexist within families. But, when coexisting in familial cases, nearly all reported cases revealed mutations of the motor neuron and pancreas homeobox 1 (MNX1) gene. Familial cases of Currarino triad combined with Mullerian duct anomaly without MNX1 gene mutation are very rare. Here we report cases of mother and daughter, who had Currarino triad and Mullerian duct anomaly without MNX1 gene mutation, along with a brief literature review.
Keyword
MeSH Terms
Figure
Reference
-
1. Currarino G, Coln D, Votteler T. Triad of anorectal, sacral, and presacral anomalies. AJR Am J Roentgenol. 1981; 137:395–398.2. Merello E, De Marco P, Ravegnani M, Riccipetitoni G, Cama A, Capra V. Novel MNX1 mutations and clinical analysis of familial and sporadic Currarino cases. Eur J Med Genet. 2013; 56:648–654.3. Cretolle C, Zerah M, Jaubert F, Sarnacki S, Revillon Y, Lyonnet S, et al. New clinical and therapeutic perspectives in Currarino syndrome (study of 29 cases). J Pediatr Surg. 2006; 41:126–131.4. Park KW, Kim JT, Kim WK. The complex of congenital caudal anomalies including anorectal malformations, sacral bony anomalities, and presacral mass: Currarino triad. J Korean Surg Soc. 1986; 31:783–788.5. Isik N, ElmaciI , Gokben B, Balak N, Tosyali N. Currarino triad: surgical management and follow-up results of four [correction of three] cases. Pediatr Neurosurg. 2010; 46:110–119.6. Markljung E, Adamovic T, Cao J, Naji H, Kaiser S, Wester T, et al. Novel mutations in the MNX1 gene in two families with Currarino syndrome and variable phenotype. Gene. 2012; 507:50–53.7. Yates VD, Wilroy RS, Whitington GL, Simmons JC. Anterior sacral defects: an autosomal dominantly inherited condition. J Pediatr. 1983; 102:239–242.8. Ross AJ, Ruiz-Perez V, Wang Y, Hagan DM, Scherer S, Lynch SA, et al. A homeobox gene, HLXB9, is the major locus for dominantly inherited sacral agenesis. Nat Genet. 1998; 20:358–361.9. Robbins JB, Broadwell C, Chow LC, Parry JP, Sadowski EA. Müllerian duct anomalies: embryological development, classification, and MRI assessment. J Magn Reson Imaging. 2015; 41:1–12.10. Ribeiro SC, Tormena RA, Peterson TV, Gonzales Mde O, Serrano PG, Almeida JA, et al. Müllerian duct anomalies: review of current management. Sao Paulo Med J. 2009; 127:92–96.