J Korean Med Sci.
2005 Aug;20(4):603-606. 10.3346/jkms.2005.20.4.603.
Investigation of the Bovine Leukemia Virus Proviral DNA in Human Leukemias and Lung cancers in Korea
- Affiliations
-
- 1Department of Clinical Pathology, The Catholic University of Korea, Medical College, Korea. hankja@catholic.ac.kr
- 2Department of General Toxicology, National Institute of Toxicological Research, Seoul, Korea.
- KMID: 1712740
- DOI: http://doi.org/10.3346/jkms.2005.20.4.603
Abstract
- The bovine leukemia virus (BLV) is the causative agent of enzootic bovine leucosis. This study investigated the presence of the BLV in leukemia (179 acute lymphoblastic leukemia, 292 acute myeloid leukemia and 46 chronic myelogenous leukemia cases) and 162 lung cancer patients (139 adenocarcinoma, 23 squamous cell carcinoma) to determine if the BLV is a causative organism of leukemia and lung cancer in Koreans. A BLV infection was confirmed in human cells by PCR using a BLV-8 primer combination. All 517 cases of human leukemia and 162 lung cancer were negative for a PCR of the BLV proviral DNA. In conclusion, although meat has been imported from BLV endemic areas, the BLV infection does not appear to be the cause of human leukemia or lung cancer in Koreans. These results can be used as a control for further studies on the BLV in Koreans.
Keyword
MeSH Terms
-
Acute Disease
Adenocarcinoma/virology
Cell Line
DNA, Viral/*genetics/isolation & purification
Humans
Korea
Leukemia/*virology
Leukemia Virus, Bovine/*genetics
Leukemia, Lymphocytic, Acute/virology
Leukemia, Myeloid/virology
Leukemia, Myeloid, Chronic/virology
Lung Neoplasms/*virology
Polymerase Chain Reaction/methods
Proviruses/*genetics
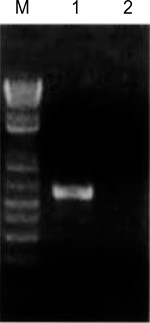
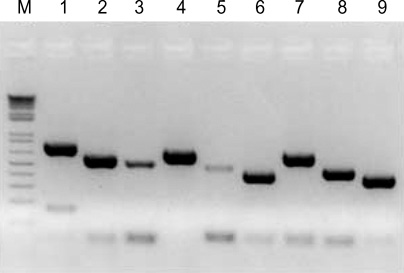
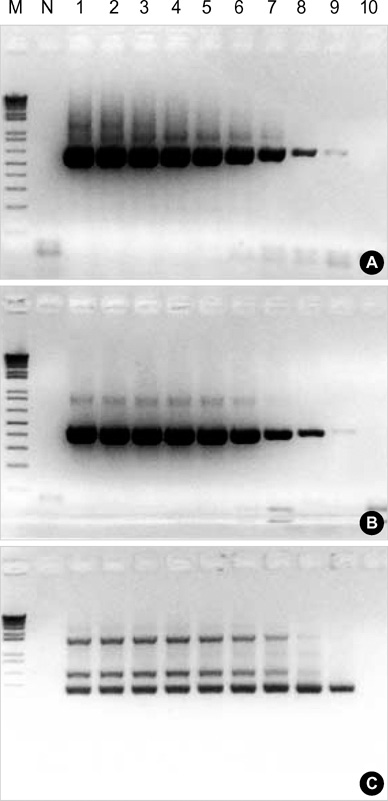
Figure
Reference
-
1. Johnson ES, Griswold CM. Oncogenic retroviruses of cattle, chickens and turkeys:potential infectivity and oncogenicity for humans. Med Hypotheses. 1996. 46:354–356.2. Johnson ES, Nicholson LG, Durack DT. Detection of antibodies to avian leucosis/sarcoma viruses (ALSV) and reticuloendotheliosis viruses (REV) in humans by ELISA. Cancer Detect Prev. 1995. 19:394–404.3. VanLeeuwen JA, Keefe GP, Tremblay R, Power C, Wichtel JJ. Seroprevalence of infection with Mycobacterium avium subspecies paratuberculosis, bovine leukemia virus, and bovine viral diarrhea virus in maritime Canada dairy cattle. Can Vet J. 2001. 42:193–198.4. Johnson ES, Fischman HR, Matanoski GM, Diamond E. Occurrence of cancer in women in the meat industry. Br J Ind Med. 1986. 43:597–604.
Article5. Johnson ES, Dalmas D, Noss J, Matanoski GM. Cancer mortality among workers in abattoirs and meatpacking plants: an update. Am J Ind Med. 1995. 27:389–403.
Article6. Rola M, Kuzmak J. The detection of bovine leukemia virus proviral DNA by PCR-ELISA. J Virol Methods. 2002. 99:33–40.
Article7. Ghysdael J, Bruck C, Kettmann R, Burny A. Bovine leukemia virus. Curr Top Microbiol Immunol. 1984. 112:1–19.
Article8. Johnson ES, Nicholson LG, Durack DT. Poultry oncogenic retroviruses and humans. Cancer Detect Prev. 1994. 18:9–31.9. McClure HM, Keeling ME, Custer RP, Marshak RR, Abt DA, Ferrer JF. Erythroleukemia in two infant chimpanzees fed milk from cows naturally infected with the bovine C-type virus. Cancer Res. 1974. 34:2745–2757.10. Pearce NE, Smith AH, Howard JK, Sheppard RA, Giles HJ, Teague CA. Non-Hodgkin's lymphoma and exposure to phenoxyherbicides, chlorophenols, fencing work, and meat works employment: a case-control study. Br J Ind Med. 1996. 43:75–83.
Article11. Johnson ES. Cancer mortality among workers in the meat department of supermarkets. Occup Environ Med. 1991. 51:541–547.
Article12. Sherman MP, Ehrlich GD, Ferrer JF, Sninsky JJ, Zandomeni R, Dock NL, Poiesz B. Amplification and analysis of specific DNA and RNA sequence of bovine leukemia virus from infected cows by polymerase chain reaction. J Clin Microbiol. 1992. 30:185–191.13. Ballagi-Pordany A, Klintevall K, Merza M, Klingeborn B, Belak S. Direct detection of bovine leukemia virus infection: practical applicability of a double polymerase chain reaction. Zentralbl Veterinarmed B. 1992. 39:69–77.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Detection of BLV Proviral DNA in Korean Native Goats Experimentally Infected with Bovine Leukemia Virus by Polymerase Chain Reaction
- Infectivity of bovine leukemia virus to Korean native goats I. antibody responses and syncytium assay for Korean native goats experimentally infected with bovine leukemia virus
- Studies on the amplification and detection of bovine leukosis proviral DNA by PCR and ECL techniques
- Genetic analysis of env and gag gene fragments of bovine leukemia virus identified in cattle from Korea
- Factors Associated with HIV-1 Proviral DNA Loads in Patients with Undetectable Plasma RNA Load