J Korean Diabetes Assoc.
2007 May;31(3):187-192. 10.4093/jkda.2007.31.3.187.
Mitochondrial Gene Therapy
- Affiliations
-
- 1Mitochondrial Research Group, Department of Internal Medicine, College of Medicine, Inje University, Korea.
- KMID: 1523006
- DOI: http://doi.org/10.4093/jkda.2007.31.3.187
Abstract
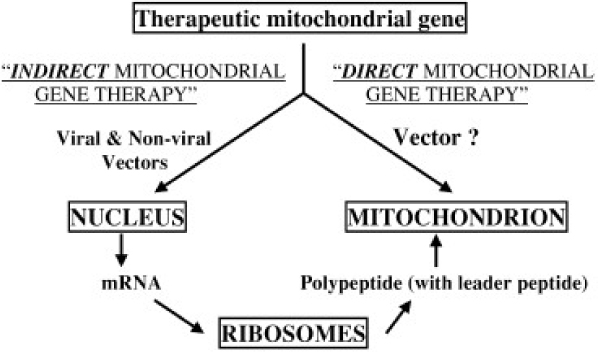
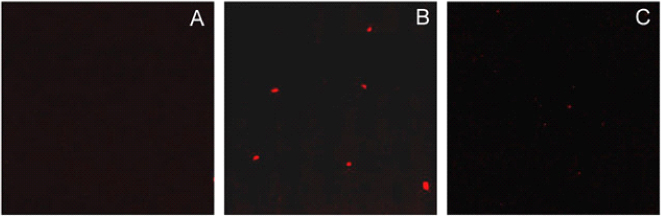
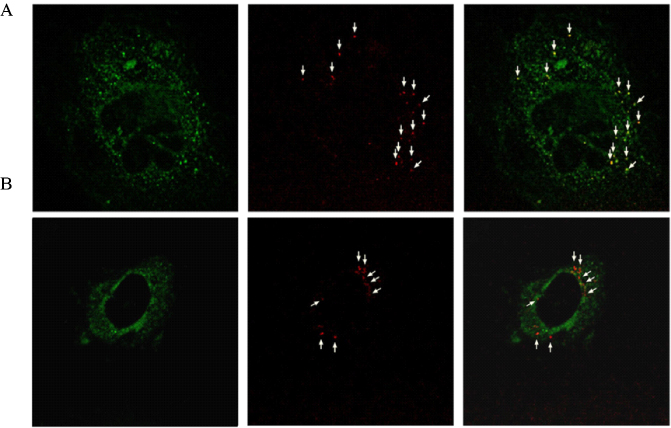
- Mitochondrial dysfunction contributes to a large variety of human disorders, ranging from neurodegenerative and neuromuscular diseases, obesity, and diabetes to ischemia-reperfusion injury and cancer. Increasing pharmacological efforts toward therapeutic interventions have been made leading to the emergence of 'Mitochondrial Medicine' as a new field of biomedical research. The identification of molecular mitochondrial drugs targets in combination with the development of methods for selectively delivering biologically active molecules to the site of mitochondria will eventually launch a multitude of new therapies for the treatment of mitochondria-related diseases, which are based either on the selective protection, repair, or eradication of cells. Yet, while tremendous efforts are being undertaken to identify new mitochondrial drugs and drug targets, the development of mitochondria-specific drug carrier systems is lagging behind. To ensure a high efficiency of current and future mitochondrial therapeutics, delivery systems need to be developed, which are able to selectively transport biologically active molecules to and into mitochondria within living cells.
MeSH Terms
Figure
Reference
-
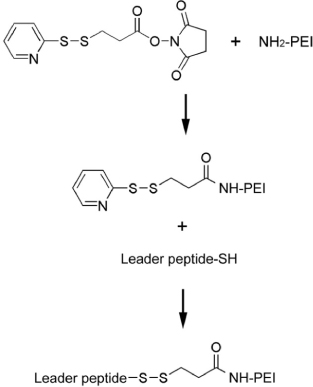
1. Luft R, Ikkos D, Palmieri G, Ernster L, Afzelius B. A case of severe hypermetabolism of nonthyroid origin with a defect in the maintenance of mitochondrial respiratory control: a correlated clinical, biochemical, and morphological study. J Clin Invest. 1962. 41:1776–1804.2. Nass MMK, Nass S. Intramitochondrial fibers with DNA characteristics. I. Fixation and electron staining reactions. J Cell Biol. 1963. 19:593–611.3. Anderson S, Bankier AT, Barell BG. Sequence and organization of the human mitochondrial genome. Nature. 1981. 290:457–465.4. Holt IJ, Harding AE, Morgan-Hughes JA. Deletions of muscle mitochondrial DNA in patients with mitochondrial myopathies. Nature. 1988. 331:717–719.5. Wallace DC, Singh G, Lott MT. Mitochondrial DNA mutations associated with Leber's hereditary optic neuropathy. Science. 1988. 242:1427–1430.6. Clayton DA. Replication and transcription of vertebrate mitochondrial DNA. Annu Rev Cell Biol. 1991. 7:453–478.7. Giles RE, Blanc H, Cann HM, Wallace DC. Maternal inheritance of human mitochondrial DNA. Proc Natl Acad Sci USA. 1980. 77:6715–6719.8. Wallace DC. Mitochondrial genetics: a paradigm for aging and degenerative disease? Science. 1992. 256:628–632.9. Modica-Napolitano JS, Singh KK. April mitochondria as targets for detection and treatment of cancer. Expert Rev Mol Med. 2002. 11:1–19.10. Chrzanowska-Lightowlers ZMA, Lightowlers RN, Turnbull DM. Gene therapy for mitochondrial DNA defects: is it possible? Gene Ther. 1995. 2:311–316.11. Barell G, Bankier AT, Drouin J. Different genetic code in human mitochondria. Nature. 1979. 282:189–194.12. Sewards R, Wiseman B, Jacobs HT. Apparent functional independence of the mitochondrial and nuclear transcription systems in cultured human cells. Mol Gen Genet. 1994. 245:760–768.13. Lee M, Choi JS, Choi MJ, Kim-Pak Y, Rhee BD, Ko KS. DNA delivery to the mitochondrial sites using mitochondrial leader peptide conjugated polyethylenimine. J Drug Targeting. 2007. 15:115–122.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Point Mutations in a Mitochondrial Transfer RiboNucleic Acid Gene in South Korean Women with Preeclampsia
- Nutrition Therapy for Mitochondrial Neurogastrointestinal Encephalopathy with Homozygous Mutation of the TYMP Gene
- MELAS Syndrome Confirmed by Mitochondrial DNA Analysis in Siblings
- Morphology and Mitochondrial Genome of Fischoederius sp. 1 in Thailand
- Species Identification Using Cytochrome B Gene