Hanyang Med Rev.
2012 Aug;32(3):154-158. 10.7599/hmr.2012.32.3.154.
Clinical Applications of Neural Stem Cells for the Treatment of Peripheral Neuropathy
- Affiliations
-
- 1Department of Neurology, Ewha Womans University School of Medicine, Seoul, Korea. bochoi@ewha.ac.kr
- KMID: 1436769
- DOI: http://doi.org/10.7599/hmr.2012.32.3.154
Abstract
- Extraordinary advances in stem cell research have initiated an era of hope for strategies to treat intractable human diseases. Personalized regenerative treatment using stem cells is expected to accelerate continuous investment and research efforts throughout the world. Despite of their constraints, adult stem cells and embryonic stem cells have been used for cell transplantation for several intractable diseases. Besides adult and embryonic stem cells, the recent studies of induced pluripotent stem cells widened the possibility of patient-specific cell therapy, drug discovery, and disease modeling. This review focuses on the developments and potential applications of the stem cells for the treatment of peripheral neuropathy.
MeSH Terms
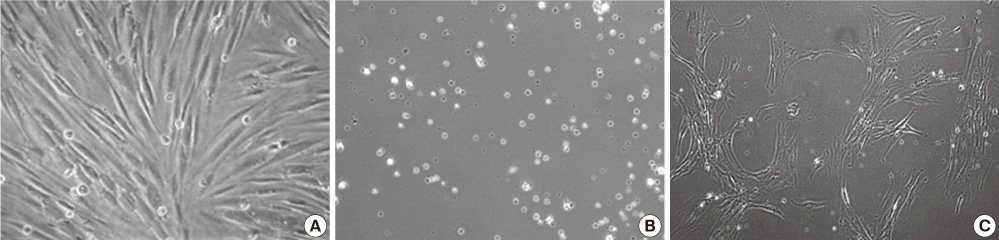
Figure
Reference
-
1. Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998. 282:1145–1147.
Article2. Mimeault M, Batra SK. Recent progress on tissue-resident adult stem cell biology and their therapeutic implications. Stem Cell Rev. 2008. 4:27–49.
Article3. Park IH, Zhao R, West JA, Yabuuchi A, Huo H, Ince TA, et al. Reprogramming of human somatic cells to pluripotency with defined factors. Nature. 2008. 451:141–146.
Article4. Shi Y, Do JT, Desponts C, Hahm HS, Scholer HR, Ding S. A combined chemical and genetic approach for the generation of induced pluripotent stem cells. Cell Stem Cell. 2008. 2:525–528.5. Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006. 126:663–676.6. Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007. 318:1917–1920.
Article7. Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007. 131:861–872.
Article8. Draper JS, Andrews PW. Embryonic stem cells: advances toward potential therapeutic use. Curr Opin Obstet Gynecol. 2002. 14:309–315.
Article9. Lerou PH, Daley GQ. Therapeutic potential of embryonic stem cells. Blood Rev. 2005. 19:321–331.
Article10. Xu C. Progress and prospects of human pluripotent stem cell research. Curr Stem Cell Res Ther. 2010. 5:205–206.
Article11. De Bari C, Kurth TB, Augello A. Mesenchymal stem cells from development to postnatal joint homeostasis, aging, and disease. Birth Defects Res C Embryo Today. 2010. 90:257–271.
Article12. McKenna DH, Brunstein CG. Umbilical cord blood: current status and future directions. Vox Sang. 2011. 100:150–162.
Article13. Rizzoti K. Adult pituitary progenitors/stem cells: from in vitro characterization to in vivo function. Eur J Neurosci. 2010. 32:2053–2062.
Article14. Fraser JK, Schreiber RE, Zuk PA, Hedrick MH. Adult stem cell therapy for the heart. Int J Biochem Cell Biol. 2004. 36:658–666.
Article15. Fukuda H, Takahashi J, Watanabe K, Hayashi H, Morizane A, Koyanagi M, et al. Fluorescence-activated cell sorting-based purification of embryonic stem cell-derived neural precursors averts tumor formation after transplantation. Stem Cells. 2006. 24:763–771.
Article16. Okita K, Ichisaka T, Yamanaka S. Generation of germline-competent induced pluripotent stem cells. Nature. 2007. 448:313–317.
Article17. Keene CD, Ortiz-Gonazalez XR, Jiang Y, Verfaillie CM, Low WC. Sanberg CD, Sanberg PR, editors. Therapeutic application of bone marrow-derived stem cells in neurologic injury and disease. Cell therapy, stem cells, and brain repair. 2006. Totowa (NJ): Humana Press;163–197.
Article18. Sanchez-Ramos J, Song S, Cardozo-Pelaez F, Hazzi C, Stedeford T, Willing A, et al. Adult bone marrow stromal cells differentiate into neural cells in vitro. Exp Neurol. 2000. 164:247–256.
Article19. Safety Study of GRNOPC1 in Spinal Cord Injury [Internet]. cited 2012 May 11. Menlo Park (CA): Geron Corporation;Available from: http://clinicaltrials.gov/ct2/show/NCT01217008?term=GRNOPC1&rank=1.
Article20. Park KI, Teng YD, Snyder EY. The injured brain interacts reciprocally with neural stem cells supported by scaffolds to reconstitute lost tissue. Nat Biotechnol. 2002. 20:1111–1117.21. Teng YD, Lavik EB, Qu X, Park KI, Ourednik J, Zurakowski D, et al. Functional recovery following traumatic spinal cord injury mediated by a unique polymer scaffold seeded with neural stem cells. Proc Natl Acad Sci U S A. 2002. 99:3024–3029.22. Ashton RS, Banerjee A, Punyani S, Schaffer DV, Kane RS. Scaffolds based on degradable alginate hydrogels and poly (lactide-co-glycolide) microspheres for stem cell culture. Biomaterials. 2007. 28:5518–5525.
Article23. Cullen DK, Stabenfeldt SE, Simon CM, Tate CC, LaPlaca MC. In vitro neural injury model for optimization of tissue-engineered constructs. J Neurosci Res. 2007. 85:3642–3651.
Article24. Tysseling-Mattiace VM, Sahni V, Niece KL, Birch D, Czeisler C, Fehlings MG, et al. Self-assembling nanofibers inhibit glial scar formation and promote axon elongation after spinal cord injury. J Neurosci. 2008. 28:3814–3823.
Article25. Pluchino S, Zanotti L, Rossi B, Brambilla E, Ottoboni L, Salani G, et al. Neurosphere-derived multipotent precursors promote neuroprotection by an immunomodulatory mechanism. Nature. 2005. 436:266–271.
Article26. Lee JP, Jeyakumar M, Gonzalez R, Takahashi H, Lee PJ, Baek RC, et al. Stem cells act through multiple mechanisms to benefit mice with neurodegenerative metabolic disease. Nat Med. 2007. 13:439–447.
Article27. Study of HuCNS-SC cells in patients with infantile or late infantile Neuronal Ceroid Lipofuscinosis (NCL) [Internet]. cited 2012 Apr 25. Bethesda (MD): U. S. National Institute of Health;Available from: http://www.clinicaltrials.gov/ct2/show/NCT00337636?term-batten&rank=4.
Article28. Wernig M, Zhao JP, Pruszak J, Hedlund E, Fu D, Soldner F, et al. Neurons derived from reprogrammed fibroblasts functionally integrate into the fetal brain and improve symptoms of rats with Parkinson's disease. Proc Natl Acad Sci U S A. 2008. 105:5856–5861.
Article29. Soldner F, Hockemeyer D, Beard C, Gao Q, Bell GW, Cook EG, et al. Parkinson's disease patient-derived induced pluripotent stem cells free of viral reprogramming factors. Cell. 2009. 136:964–977.
Article30. Ebert AD, Yu J, Rose FF Jr, Mattis VB, Lorson CL, Thomson JA, et al. Induced pluripotent stem cells from a spinal muscular atrophy patient. Nature. 2009. 457:277–280.
Article31. Lee G, Papapetrou EP, Kim H, Chambers SM, Tomishima MJ, Fasano CA, et al. Modelling pathogenesis and treatment of familial dysautonomia using patient-specific iPSCs. Nature. 2009. 461:402–406.32. Carvajal-Vergara X, Sevilla A, D'Souza SL, Ang YS, Schaniel C, Lee DF, et al. Patient-specific induced pluripotent stem-cell-derived models of LEOPARD syndrome. Nature. 2010. 465:808–812.
Article33. Hanna J, Wernig M, Markoulaki S, Sun CW, Meissner A, Cassady JP, et al. Treatment of sickle cell anemia mouse model with iPS cells generated from autologous skin. Science. 2007. 318:1920–1923.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Adult Neurogenesis in the Central and Peripheral Nervous Systems
- Regulation of Neural Stem Cell Fate by Natural Products
- Clinical and Electrophysiologic Characteristics of Paraproteinemic Neuropathy
- Introduction to the Management and Diagnosis of Peripheral Neuropathies
- Treatment of peripheral neuropathy: a multidisciplinary approach is necessary