J Vet Sci.
2007 Mar;8(1):45-49. 10.4142/jvs.2007.8.1.45.
Sero-survey on Aino, Akabane, Chuzan, bovine ephemeral fever and Japanese encephalitis virus of cattle and swine in Korea
- Affiliations
-
- 1National Veterinary Research and Quarantine Service, Ministry of Agriculture and Forestry, Anyang 430-824, Korea. kweonch@nvrqs.go.kr
- KMID: 1104235
- DOI: http://doi.org/10.4142/jvs.2007.8.1.45
Abstract
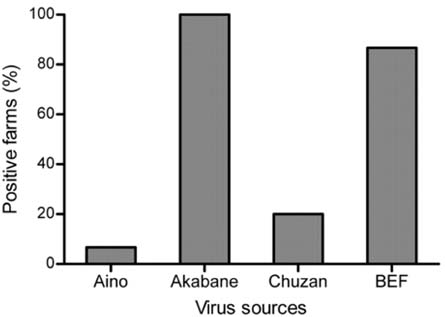
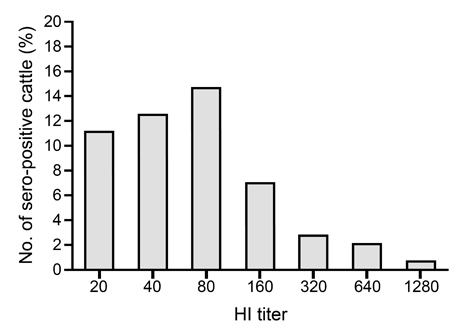
- Vector-borne arboviruses produce mild to severe symptoms in domestic animals. Bovine ephemeral fever (BEF), Akabane, Aino, and Chuzan virus have been primarily attributed to reproductive disorders or febrile diseases in cattle, and Japanese encephalitis virus (JEV) is mainly associated with reproductive failures in swine. We investigated antibody titers from domestic swine against four bovine arboviruses (BEF, Akabane, Aino, and Chuzan virus) and from cattle against JEV in Korea. While the positive rates for Akabane and BEF were 37.4% and 15.7%, the positive incidence of Chuzan and Aino were relatively low, with positive rates of 3.04% and 0.4%, respectively, based on a virus neutralization assay. Antibody titers against more than one virus were also frequently detected in domestic swine. The incidence of JEV was 51.3% among domestic cattle. In addition, one positive case was detected in the thoracic fluids from 35 aborted calves, based on the hemagglutination inhibition test. Our results indicate that swine are susceptible hosts of bovine arboviruses without showing clinical symptoms in a natural environment. Moreover, we confirmed that JEV could be associated with reproductive failure in pregnant cattle, as were other vector-borne bovine arboviruses assessed in this study.
Keyword
MeSH Terms
-
Animals
Antibodies, Viral/blood
Cattle
Cattle Diseases/*epidemiology/*virology
Encephalitis Virus, Japanese/*immunology
Encephalitis, Japanese/blood/epidemiology/*veterinary/virology
Ephemeral Fever/blood/*epidemiology/virology
Ephemeral Fever Virus, Bovine/*immunology
Hemagglutination Tests
Incidence
Korea/epidemiology
Neutralization Tests
Swine
Swine Diseases/*epidemiology/*virology
Figure
Cited by 2 articles
-
Detection of Neutralizing Antibody Against Japanese Encephalitis Virus in Wild Boars of Korea
Dong-Kun Yang, Ha-Hyun Kim, Bang-Hun Hyun, Seong-In Lim, Yun-Kyoung Nam, Jin-Ju Nah, Jae-Young Song
J Bacteriol Virol. 2012;42(4):353-356. doi: 10.4167/jbv.2012.42.4.353.Serosurveillance for Japanese encephalitis, Akabane, and Aino viruses for Thoroughbred horses in Korea
Dong-Kun Yang, Byoung-han Kim, Chang-Hee Kweon, Jin-Ju Nah, Hyun-Joo Kim, Kyung-Woo Lee, Young-Jin Yang, Kyu-Whan Mun
J Vet Sci. 2008;9(4):381-385. doi: 10.4142/jvs.2008.9.4.381.
Reference
-
1. Akashi H, Kaku Y, Kong X, Pang H. Antigenic and genetic comparisons of Japanese and Australian Simbu serogroup viruses: evidence for the recovery of natural virus reassortants. Virus Res. 1997. 50:205–213.
Article2. Akashi H, Kaku Y, Kong XG, Pang H. Sequence determination and phylogenetic analysis of the Akabane bunyavirus S RNA genome segment. J Gen Virol. 1997. 78:2847–2851.
Article3. Akashi H, Onuma S, Nagano H, Ohta M, Fukutomi T. Detection and differentiation of Aino and Akabane Simbu serogorup bunyaviruses by nested polymerase chain reaction. Arch Virol. 1999. 144:2101–2109.
Article4. Clarke DH, Casals J. Techniques for hemagglutination and hemagglutination-inhibition with arthropod-borne viruses. Am J Trop Med Hyg. 1958. 7:561–573.
Article5. Horimoto M, Sakai T, Goto H. Changes in antibody titers in cattle with Japanese encephalitis virus infection. Indian J Med Res. 1987. 86:695–701.6. Huang CC, Huang TS, Deng MC, Jong MH, Lin SY. Natural infections of pigs with akabane virus. Vet Microbiol. 2003. 94:1–11.
Article7. Ilkal MA, Dhanda V, Rao BU, George S, Mishra AC, Prasanna Y, Gopalkrishna S, Pavri KM. Absence of viraemia in cattle after experimental infection with Japanese encephalitis virus. Trans R Soc Trop Med Hyg. 1988. 82:628–631.
Article8. Johnson AJ, Martin DA, Karabatsos N, Roehrig JT. Detection of anti-arboviral immunoglobulin G by using a monoclonal antibody-based capture enzyme-linked immunosorbent assay. J Clin Microbiol. 2000. 38:1827–1831.
Article9. Kitano Y, Yamashita S, Makinoda K. A congenital abnormality of calves, suggestive of a new type of arthropod-borne virus infection. J Comp Pathol. 1994. 111:427–437.
Article10. Konno S, Nakagawa M. Akabane disease in cattle: congenital abnormalities casued by viral infection. Experimental disease. Vet Pathol. 1982. 19:267–279.
Article11. Konno S, Moriwaki M, Nakagawa M. Akabane disease in cattle: congenital abnormalities caused by viral infection. Spontaneous disease. Vet Pathol. 1982. 19:246–266.
Article12. Ma SP, Yoshida Y, Makino Y, Tadano M, Ono T, Ogawa M. Short report: a major genotype of Japanese encephalitis currently circulating in Japan. Am J Trop Med Hyg. 2003. 69:151–154.
Article13. Morimoto T, Kurogi H, Miura Y, Sugimori T, Fujisaki Y. Isolation of Japanese encephalitis virus and a hemagglutinating DNA virus from the brain of stillborn piglets. Natl Inst Anim Health Q (Tokyo). 1972. 12:127–136.14. Nandi S, Negi BS. Bovine ephemeral fever: a review. Comp Immunol Microbiol Infect Dis. 1999. 22:81–91.
Article15. Norton JH, Tranter WP, Campbell RS. A farming systems study of abortion in dairy cattle on the Atherton Tableland. 2. The pattern of infectious disease. Aust Vet J. 1989. 66:163–167.
Article16. Ohashi S, Matsumori Y, Yanase T, Yamakawa M, Kato T, Tsuda T. Evidence of an antigenic shift among Palyam serogroup orbiviruses. J Clin Microbiol. 2004. 42:4610–4614.
Article17. Olson KE, Adelman ZN, Travanty EA, Sanchez-Vargas I, Beaty BJ, Blair CD. Developing arbovirus resistance in mosquitoes. Insect Biochem Mol Biol. 2002. 32:1333–1343.
Article18. Rajendran R, Thenmozhi V, Tewari SC, Balasubramanian A, Ayanar K, Manavalan R, Gajanana A, Kabilan L, Thakare JP, Satyanarayana K. Longitudinal studies in South Indian villages on Japanese encephalitis virus infection in mosquitoes and seroconversion in goats. Trop Med Int Health. 2003. 8:174–181.
Article19. Reuben R, Thenmozhi V, Samuel PP, Gajanana A, Mani TR. Mosquito blood feeding patterns as a factor in the epidemiology of Japanese encephalitis in southern India. Am J Trop Med Hyg. 1992. 46:654–663.
Article20. Solomon T, Ni H, Beasley DW, Ekkelenkamp M, Cardosa MJ, Barrett AD. Origin and evolution of Japanese encephalitis virus in southeast Asia. J Virol. 2003. 77:3091–3098.
Article21. Uren MF, St George TD, Murphy GM. Studies on the pathogenesis of bovine ephemeral fever in experimental cattle III. Virological and biochemical data. Vet Microbiol. 1992. 30:297–307.
Article22. van den Heuvel JF, Hogenhout SA, van der Wilk F. Recognition and receptors in virus transmission by arthropods. Trends Microbiol. 1999. 7:71–76.
Article23. Weber F, Elliott RM. Antigenic drift, antigenic shift and interferon antagonists: how bunyaviruses counteract the immune system. Virus Res. 2002. 88:129–136.
Article24. Wu SC, Lin CW, Lee SC, Lian WC. Phenotypic and genotypic characterization of the neurovirulence and neuroinvasiveness of a large-plaque attenuated Japanese encephalitis virus isolate. Microbes Infect. 2003. 5:475–480.
Article25. Yamakawa M, Furuuchi S, Minobe Y. Molecular characterization of double-stranded RNA segments encoding the major capsid proteins of a Palyam serogroup orbivirus that caused an epizootic of congenital abnormalities in cattle. J Gen Virol. 1999. 80:205–208.
Article26. Yanase T, Kato T, Kubo T, Yoshida K, Ohashi S, Yamakawa M, Miura Y, Tsuda T. Isolation of bovine arboviruses from Culicoides biting midges (Diptera: Ceratopogonidae) in southern Japan: 1985-2002. J Med Entomol. 2005. 42:63–67.
Article27. Yeruham I, Sharir B, Yadin H, Tiomkin D, Chai D. Bovine ephemeral fever in beef cattle herds in the Jordan Valley, Israel. Vet Rec. 2003. 152:86–88.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Monitoring of Five Bovine Arboviral Diseases Transmitted by Arthropod Vectors in Korea
- Development of inactivated Akabane and bovine ephemeral fever vaccine for cattle
- Apoptosis in Vero cells infected with Akabane, Aino and Chuzan virus
- Analysis of Five Arboviruses and Culicoides Distribution on Cattle Farms in Jeollabuk-do, Korea
- Antiviral effect of 18-mer-peptide (1b-4/21-C12) on Japanese encephalitis virus and Akabane virus