Korean J Lab Med.
2009 Aug;29(4):331-337. 10.3343/kjlm.2009.29.4.331.
Comparison of Quantitative Results among Two Automated Rapid Plasma Reagin (RPR) Assays and a Manual RPR Test
- Affiliations
-
- 1Department of Laboratory Medicine, College of Medicine, The Catholic University of Korea, Seoul, Korea. vsykim@catholic.ac.kr
- 2Department of Pathology, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- KMID: 1096927
- DOI: http://doi.org/10.3343/kjlm.2009.29.4.331
Abstract
-
BACKGROUND: We compared two automated Rapid Plasma Reagin (RPR) assay kits with a manual RPR assay kit to evaluate the possibility of using the two automated RPR assays as an alternative to the manual RPR assay for a quantitative monitoring.
METHODS
One hundred eighty-five samples were analyzed, including 16 sera from patients with primary, secondary, and latent syphilis. Measured RPR unit (R.U.) values of two automated RPR assay kits, Mediace RPR (Sekisui Chemical Co., Ltd, Japan) and HBi Auto RPR (HBI Co., Ltd, Korea), were compared with the RPR titers of Macro-Vue RPR card test (Becton Dickinson BD Microbiology systems, USA). As a confirmatory test, Anti-Treponema pallidum EUROLINE WB (IgG) and Anti-Treponema pallidum EUROLINE WB (IgM) (Euroimmun, Germany) were used.
RESULTS
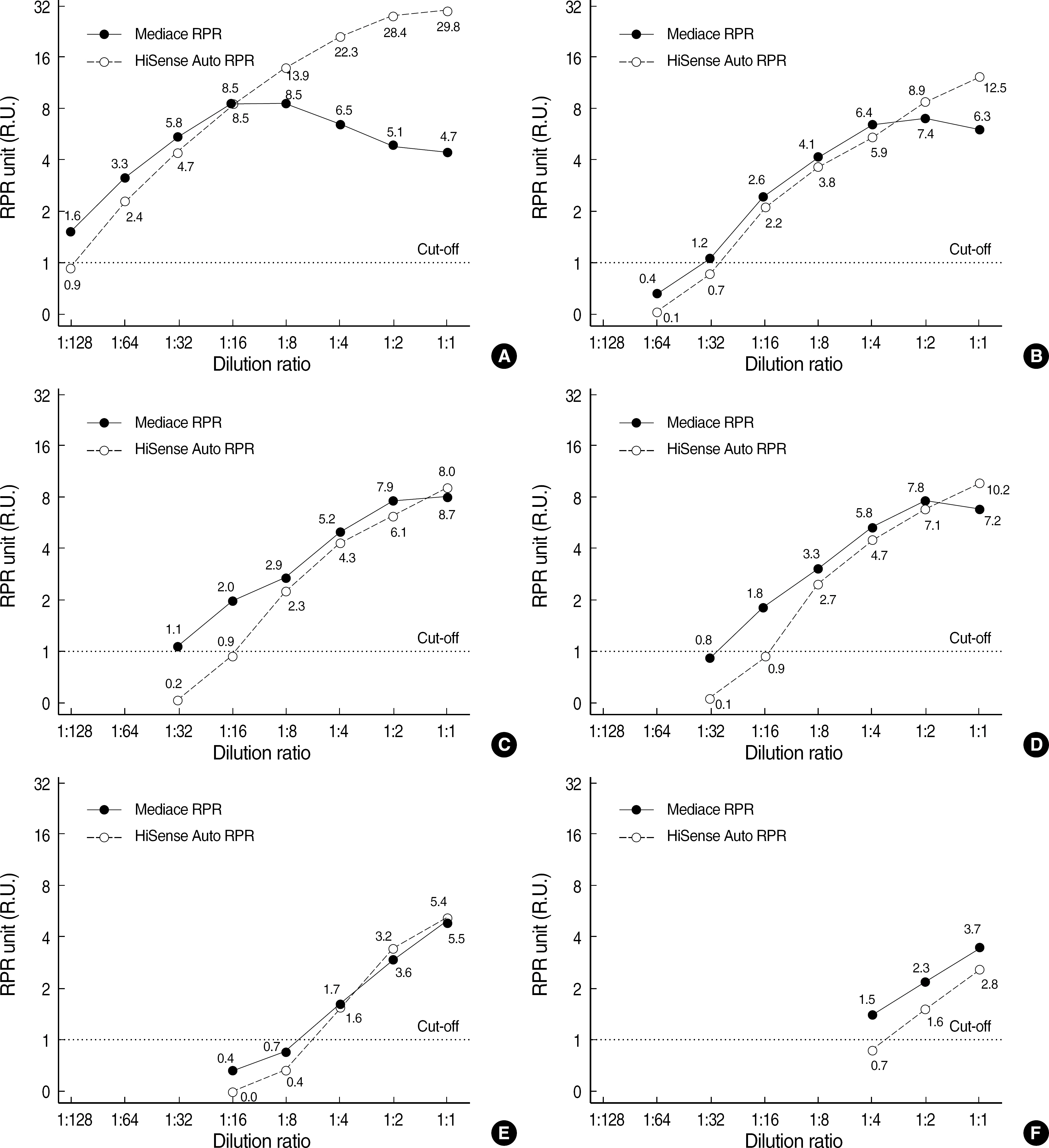
There was a prozone effect with Mediace RPR at RPR titer (card test) of 1:16, but not with HBi Auto RPR. The R.U. values of the two automated RPR assays did not show proportional increase to the RPR titer. Agreement between manual RPR and two automated RPR assay kits, Mediace RPR assay and HBi Auto RPR assay, were 83.8% and 83.2%, respectively.
CONCLUSIONS
The two automated RPR assay kits could not be used as an alternative to manual RPR test for quantitative analysis of RPR titer. As Mediace RPR shows a prozone effect at relatively low RPR titer, caution is needed in the interpretation of the measured values.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Comparison of Automated Treponemal and Nontreponemal Test Algorithms as First-Line Syphilis Screening Assays
Hee Jin Huh, Jae-Woo Chung, Seong Yeon Park, Seok Lae Chae
Ann Lab Med. 2016;36(1):23-27. doi: 10.3343/alm.2016.36.1.23.
Reference
-
1.Noh J., Ko HH., Yun Y., Choi YS., Lee SG., Shin S, et al. Evaluation of performance and false positivity of Mediace RPR test that uses a chemistry autoanalyzer. Korean J Lab Med. 2008. 28:312–8. (노재광, 고학 현, 윤여민, 최영숙, 이상곤, 신수 등. 생화학 자동분석기용 Mediace RPR 검사의 성능 및 위양성 평가. 대한진단검사의학회지 2008;28:312-8.).
Article2.Huh HJ., Lee KK., Kim ES., Chae SL. Analysis of positive results in Mediace Rapid Plasma Reagin and Treponema pallidum Latex Agglutination as the automated syphilis test. Korean J Lab Med. 2007. 27:324–9. (허희진, 이교관, 김의석, 채석래. 자동화 매독검사인 Mediace Rapid Plasma Reagin 과 Treponema pallidum Latex Agglutination 양성결과분석. 대한진단검사의학회지 2007;27:324-9.).3.Kim J., Kim WH., Cho C., Kim J., Kim GY., Nam MH, et al. Evaluation of automated architect syphilis TP as a diagnostic laboratory screening test for syphilis. Korean J Lab Med. 2008. 28:475–82. ((김지용, 김우 현, 조치현, 김주연, 김가영, 남명현 등. 매독선별검사로서의 Architect Syphilis TP 평가. 대한진단검사의학회지 2008;28:475-82.).
Article4.McMillan A., Young H. Qualitative and quantitative aspects of the serological diagnosis of early syphilis. Int J STD AIDS. 2008. 19:620–4.
Article5.Dicker LW., Mosure DJ., Steece R., Stone KM. Laboratory tests used in US public health laboratories for sexually transmitted diseases, 2000. Sex Transm Dis. 2004. 31:259–64.
Article6.Lewis DA., Young H. Syphilis. Sex Transm Infect. 2006. 82(S4):iv. 13–5.
Article7.Young H. Syphilis. Sex Transm Dis. 1998. 16:691–8.8.Orum O., Nielsen JR., Birch-Andersen A. The effect of cholesterol on the morphology and reactivity of the mixture of lipids used in syphilis serology. APMIS. 1990. 98:9–18.9.Castro AR., Morrill WE., Shaw WA., Gale DC., Park MM., Peregrino-Ferreira LA, et al. Use of synthetic cardiolipin and lecithin in the antigen used by the venereal disease research laboratory test for serodiagnosis of syphilis. Clin Diagn Lab Immunol. 2000. 7:658–61.
Article10.Castro R., Prieto ES., Santo I., Azevedo J., Exposto Fda L. Evaluation of an enzyme immunoassay technique for detection of antibodies against Treponema pallidum. J Clin Microbiol. 2003. 41:250–3.11.Smikle MF., James OB., Prabhaker P. Biological false positive serological tests for syphilis in the Jamaican population. Genitourin Med. 1990. 66:76–8.
Article12.Geusau A., Kittler H., Hein U., Dangl-Erlach E., Stingl G., Tschachler E. Biological false-positive tests comprise a high proportion of Venereal Disease Research Laboratory reactions in an analysis of 300,000 sera. Int J STD AIDS. 2005. 16:722–6.
Article13.Lee CY., Han SM., Kang KC. Evaluation of Treponema pallidum antibody test in blood donors. Korean J Blood Transfus. 1997. 8:263–9. (이충영, 한상무, 강권철. 공혈자에서매독균항체검사의비교평가. 대한수혈학회지 1997;8:263-9.).
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Practical Application of Quantitative HiSens Auto Rapid Plasma Reagin Latex Turbidimetric Immunoagglutination for Diagnosing Syphilis; Comparison Analysis between Rapid Plasma Reagin Latex Turbidimetric Immunoagglutination Test and Rapid Plasma Reagin Card Test
- Evaluation of AutoLab Rapid Plasma Reagin and AutoLab Treponema pallidum Latex Agglutination for Syphilis Infection Testing
- Analysis of Positive Results in Mediace Rapid Plasma Reagin and Treponema pallidum Latex Agglutination as the Automated Syphilis Test
- Analysis of Positive Results in Mediace Rapid Plasma Reagin and Treponema pallidum Latex Agglutination at a University Hospital
- Serologic Test for Syphilis by Mediace RPR Test for Chemistry Autoanalyzer