Korean J Physiol Pharmacol.
2017 Jan;21(1):19-26. 10.4196/kjpp.2017.21.1.19.
Betulin suppressed interleukin-1β-induced gene expression, secretion and proteolytic activity of matrix metalloproteinase in cultured articular chondrocytes and production of matrix metalloproteinase in the knee joint of rat
- Affiliations
-
- 1Department of Orthopedic Surgery, Gangneung Asan Hospital, College of Medicine, University of Ulsan, Gangneung 25440, Korea.
- 2Department of Health Management, Sahmyook University, Seoul 01795, Korea.
- 3Department of Orthopedic Surgery and Institute of Health Sciences, School of Medicine and Hospital, Gyeongsang National University, Jinju 52727, Korea. LCJ123@cnu.ac.kr
- 4Department of Pharmacology, School of Medicine, Chungnam National University, Daejeon 35015, Korea. hscspine@hanmail.net
- KMID: 2371064
- DOI: http://doi.org/10.4196/kjpp.2017.21.1.19
Abstract
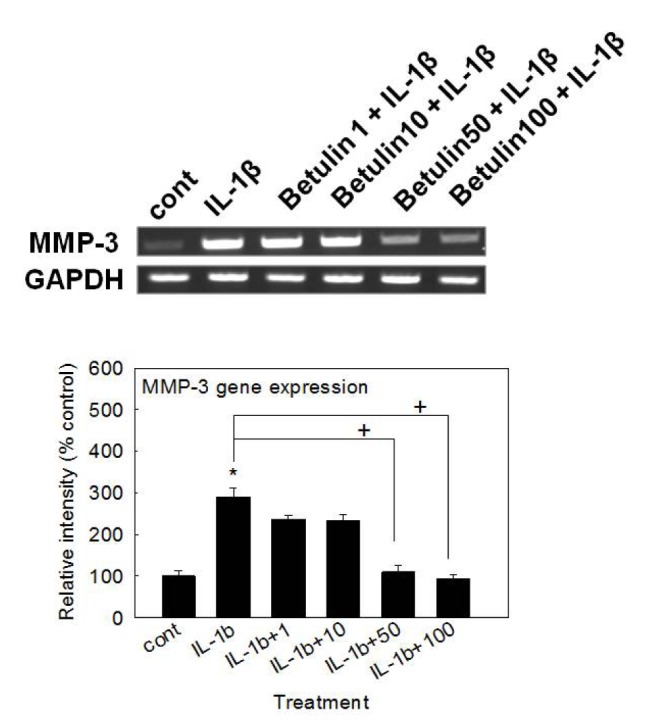
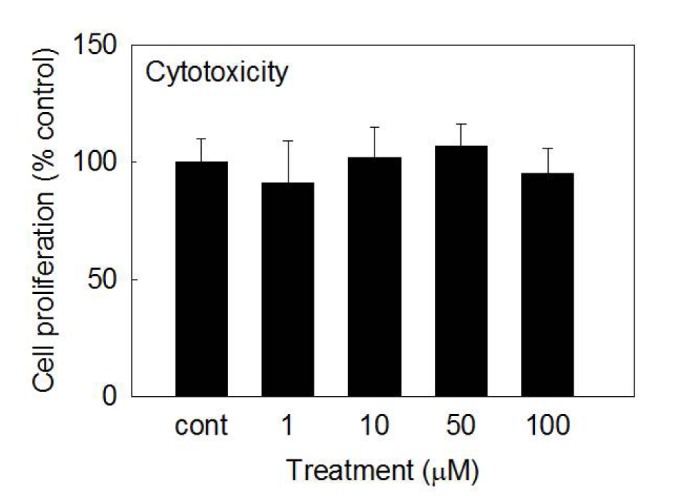
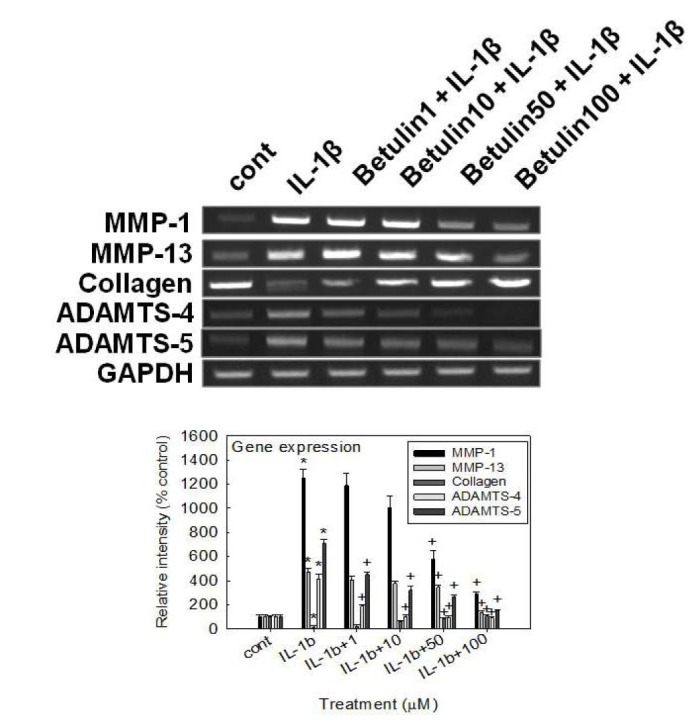
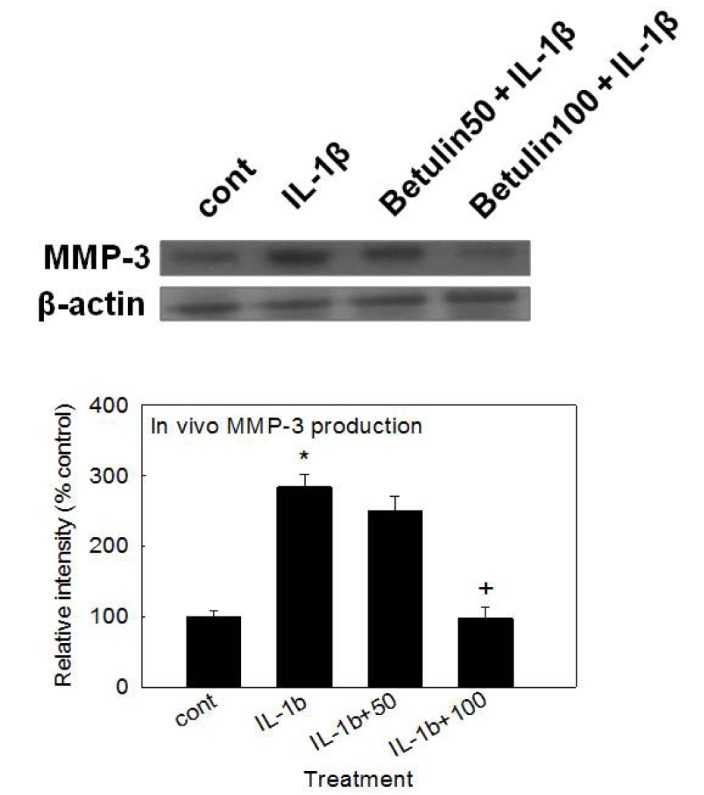
- We investigated whether betulin affects the gene expression, secretion and proteolytic activity of matrix metalloproteinase-3 (MMP-3) in primary cultured rabbit articular chondrocytes, as well as in vivo production of MMP-3 in the rat knee joint to evaluate the potential chondroprotective effect of betulin. Rabbit articular chondrocytes were cultured and reverse transcription-polymerase chain reaction (RT-PCR) was used to measure interleukin-1β (IL-1β)-induced gene expression of MMP-3, MMP-1, MMP-13, a disintegrin and metalloproteinase with thrombospondin motifs-4 (ADAMTS-4), ADAMTS-5 and type II collagen. Effect of betulin on IL-1β-induced secretion and proteolytic activity of MMP-3 was investigated using western blot analysis and casein zymography, respectively. Effect of betulin on MMP-3 protein production was also examined in vivo. The results were as follows: (1) betulin inhibited the gene expression of MMP-3, MMP-1, MMP-13, ADAMTS-4, and ADAMTS-5, but increased the gene expression of type II collagen; (2) betulin inhibited the secretion and proteolytic activity of MMP-3; (3) betulin suppressed the production of MMP-3 protein in vivo. These results suggest that betulin can regulate the gene expression, secretion, and proteolytic activity of MMP-3, by directly acting on articular chondrocytes.
Keyword
MeSH Terms
Figure
Reference
-
1. Aigner T, McKenna L. Molecular pathology and pathobiology of osteoarthritic cartilage. Cell Mol Life Sci. 2002; 59:5–18. PMID: 11846033.
Article2. Mankin HJ. The response of articular cartilage to mechanical injury. J Bone Joint Surg Am. 1982; 64:460–466. PMID: 6174527.
Article3. Dean DD, Martel-Pelletier J, Pelletier JP, Howell DS, Woessner JF Jr. Evidence for metalloproteinase and metalloproteinase inhibitor imbalance in human osteoarthritic cartilage. J Clin Invest. 1989; 84:678–685. PMID: 2760206.
Article4. Kullich W, Fagerer N, Schwann H. Effect of the NSAID nimesulide on the radical scavenger glutathione S-transferase in patients with osteoarthritis of the knee. Curr Med Res Opin. 2007; 23:1981–1986. PMID: 17631696.
Article5. Birkedal-Hansen H, Moore WG, Bodden MK, Windsor LJ, Birkedal-Hansen B, DeCarlo A, Engler JA. Matrix metalloproteinases: a review. Crit Rev Oral Biol Med. 1993; 4:197–250. PMID: 8435466.
Article6. Burrage PS, Mix KS, Brinckerhoff CE. Matrix metalloproteinases: role in arthritis. Front Biosci. 2006; 11:529–543. PMID: 16146751.
Article7. Garnero P, Rousseau JC, Delmas PD. Molecular basis and clinical use of biochemical markers of bone, cartilage, and synovium in joint diseases. Arthritis Rheum. 2000; 43:953–968. PMID: 10817547.
Article8. Lin PM, Chen CT, Torzilli PA. Increased stromelysin-1 (MMP-3), proteoglycan degradation (3B3- and 7D4) and collagen damage in cyclically load-injured articular cartilage. Osteoarthritis Cartilage. 2004; 12:485–496. PMID: 15135145.
Article9. Freemont AJ, Hampson V, Tilman R, Goupille P, Taiwo Y, Hoyland JA. Gene expression of matrix metalloproteinases 1, 3, and 9 by chondrocytes in osteoarthritic human knee articular cartilage is zone and grade specific. Ann Rheum Dis. 1997; 56:542–549. PMID: 9370879.
Article10. Goupille P, Jayson MI, Valat JP, Freemont AJ. Matrix metalloproteinases: the clue to intervertebral disc degeneration? Spine (Phila Pa 1976). 1998; 23:1612–1626. PMID: 9682320.
Article11. Kanyama M, Kuboki T, Kojima S, Fujisawa T, Hattori T, Takigawa M, Yamashita A. Matrix metalloproteinases and tissue inhibitors of metalloproteinases in synovial fluids of patients with temporomandibular joint osteoarthritis. J Orofac Pain. 2000; 14:20–30. PMID: 11203734.12. Jo H, Park JS, Kim EM, Jung MY, Lee SH, Seong SC, Park SC, Kim HJ, Lee MC. The in vitro effects of dehydroepiandrosterone on human osteoarthritic chondrocytes. Osteoarthritis Cartilage. 2003; 11:585–594. PMID: 12880581.
Article13. Little CB, Barai A, Burkhardt D, Smith SM, Fosang AJ, Werb Z, Shah M, Thompson EW. Matrix metalloproteinase 13-deficient mice are resistant to osteoarthritic cartilage erosion but not chondrocyte hypertrophy or osteophyte development. Arthritis Rheum. 2009; 60:3723–3733. PMID: 19950295.
Article14. Neuhold LA, Killar L, Zhao W, Sung ML, Warner L, Kulik J, Turner J, Wu W, Billinghurst C, Meijers T, Poole AR, Babij P, DeGennaro LJ. Postnatal expression in hyaline cartilage of constitutively active human collagenase-3 (MMP-13) induces osteoarthritis in mice. J Clin Invest. 2001; 107:35–44. PMID: 11134178.
Article15. Yoshihara Y, Nakamura H, Obata K, Yamada H, Hayakawa T, Fujikawa K, Okada Y. Matrix metalloproteinases and tissue inhibitors of metalloproteinases in synovial fluids from patients with rheumatoid arthritis or osteoarthritis. Ann Rheum Dis. 2000; 59:455–461. PMID: 10834863.
Article16. Echtermeyer F, Bertrand J, Dreier R, Meinecke I, Neugebauer K, Fuerst M, Lee YJ, Song YW, Herzog C, Theilmeier G, Pap T. Syndecan-4 regulates ADAMTS-5 activation and cartilage breakdown in osteoarthritis. Nat Med. 2009; 15:1072–1076. PMID: 19684582.
Article17. Stanton H, Rogerson FM, East CJ, Golub SB, Lawlor KE, Meeker CT, Little CB, Last K, Farmer PJ, Campbell IK, Fourie AM, Fosang AJ. ADAMTS5 is the major aggrecanase in mouse cartilage in vivo and in vitro. Nature. 2005; 434:648–652. PMID: 15800625.
Article18. Nam DC, Kim BK, Lee HJ, Shin HD, Lee CJ, Hwang SC. Effects of prunetin on the proteolytic activity, secretion and gene expression of MMP-3 in vitro and production of MMP-3 in vivo. KoreanJ Physiol Pharmacol. 2016; 20:221–228. PMID: 26937219.19. Park JS, Kim DK, Shin HD, Lee HJ, Jo HS, Jeong JH, Choi YL, Lee CJ, Hwang SC. Apigenin regulates interleukin-1b-induced production of matrix metalloproteinase both in the knee joint of rat and in primary cultured articular chondrocytes. Biomol Ther (Seoul). 2016; 24:163–170. PMID: 26902085.20. Park JS, Lee HJ, Lee DY, Jo HS, Jeong JH, Kim DH, Nam DC, Lee CJ, Hwang SC. Chondroprotective effects of wogonin in experimental models of osteoarthritis in vitro and in vivo. Biomol Ther (Seoul). 2015; 23:442–448. PMID: 26336584.
Article21. Kang BJ, Ryu J, Lee CJ, Hwang SC. Luteolin inhibits the activity, secretion and gene expression of MMP-3 in cultured articular chondrocytes and production of MMP-3 in the rat knee. Biomol Ther (Seoul). 2014; 22:239–245. PMID: 25009705.
Article22. Bai T, Yang Y, Yao YL, Sun P, Lian LH, Wu YL, Nan JX. Betulin alleviated ethanol-induced alcoholic liver injury via SIRT1/AMPK signaling pathway. Pharmacol Res. 2016; 105:1–12. PMID: 26776965.
Article23. Zhao H, Zheng Q, Hu X, Shen H, Li F. Betulin attenuates kidney injury in septic rats through inhibiting TLR4/NF-kB signaling pathway. Life Sci. 2016; 144:185–193. PMID: 26656467.24. Zhao H, Liu Z, Liu W, Han X, Zhao M. Betulin attenuates lung and liver injuries in sepsis. Int Immunopharmacol. 2016; 30:50–56. PMID: 26644168.
Article25. Guo MY, Li WY, Zhang Z, Qiu C, Li C, Deng G. Betulin suppresses S. aureus-induced mammary gland inflammatory injury by regulating PPAR-γ in mice. Int Immunopharmacol. 2015; 29:824–831. PMID: 26344430.26. Zhang SY, Zhao QF, Fang NN, Yu JG. Betulin inhibits pro-inflammatory cytokines expression through activation STAT3 signaling pathway in human cardiac cells. Eur Rev Med Pharmacol Sci. 2015; 19:455–460. PMID: 25720718.27. Moon PD, Jeong HS, Chun CS, Kim HM. Baekjeolyusin-tang and its active component berberine block the release of collagen and proteoglycan from IL-1β-stimulated rabbit cartilage and down-regulate matrix metalloproteinases in rabbit chondrocytes. Phytother Res. 2011; 25:844–850. PMID: 21089182.
Article28. Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Vistica D, Warren JT, Bokesch H, Kenney S, Boyd MR. New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst. 1990; 82:1107–1112. PMID: 2359136.
Article29. Bonnet CS, Walsh DA. Osteoarthritis, angiogenesis and inflammation. Rheumatology (Oxford). 2005; 44:7–16. PMID: 15292527.
Article30. Goldring MB, Otero M, Tsuchimochi K, Ijiri K, Li Y. Defining the roles of inflammatory and anabolic cytokines in cartilage metabolism. Ann Rheum Dis. 2008; 67(Suppl 3):iii75–iii82. PMID: 19022820.
Article31. Kobayashi M, Squires GR, Mousa A, Tanzer M, Zukor DJ, Antoniou J, Feige U, Poole AR. Role of interleukin-1 and tumor necrosis factor alpha in matrix degradation of human osteoarthritic cartilage. Arthritis Rheum. 2005; 52:128–135. PMID: 15641080.32. Loeser RF. Molecular mechanisms of cartilage destruction: mechanics, inflammatory mediators, and aging collide. Arthritis Rheum. 2006; 54:1357–1360. PMID: 16645963.
Article33. Aida Y, Maeno M, Suzuki N, Shiratsuchi H, Motohashi M, Matsumura H. The effect of IL-1beta on the expression of matrix metalloproteinases and tissue inhibitors of matrix metalloproteinases in human chondrocytes. Life Sci. 2005; 77:3210–3221. PMID: 15979654.34. Pantsulaia I, Kalichman L, Kobyliansky E. Association between radiographic hand osteoarthritis and RANKL, OPG and inflammatory markers. Osteoarthritis Cartilage. 2010; 18:1448–1453. PMID: 20633673.
Article35. Lijnen HR. Matrix metalloproteinases and cellular fibrinolytic activity. Biochemistry (Mosc). 2002; 67:92–98. PMID: 11841344.36. Jeong JW, Lee HH, Choi EO, Lee KW, Kim KY, Kim SG, Hong SH, Kim GY, Park C, Kim HK, Choi YW, Choi YH. Schisandrae fructus inhibits IL-1b-induced matrix metalloproteinases and inflammatory mediators production in SW1353 human chondrocytes by suppressing NF-κB and MAPK activation. Drug Dev Res. 2015; 76:474–483. PMID: 26443270.37. Piao T, Ma Z, Li X, Liu J. Taraxasterol inhibits IL-1β-induced inflammatory response in human osteoarthritic chondrocytes. Eur J Pharmacol. 2015; 756:38–42. PMID: 25797286.
Article38. Lu S, Xiao X, Cheng M. Matrine inhibits IL-1β-induced expression of matrix metalloproteinases by suppressing the activation of MAPK and NF-κB in human chondrocytes in vitro. Int J Clin Exp Pathol. 2015; 8:4764–4772. PMID: 26191166.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Apigenin Regulates Interleukin-1β-Induced Production of Matrix Metalloproteinase Both in the Knee Joint of Rat and in Primary Cultured Articular Chondrocytes
- Effect of oleanolic acid on the activity, secretion and gene expression of matrix metalloproteinase-3 in articular chondrocytes in vitro and the production of matrix metalloproteinase-3 in vivo
- Effects of prunetin on the proteolytic activity, secretion and gene expression of MMP-3 in vitro and production of MMP-3 in vivo
- Erratum to: Betulin suppressed interleukin-1b-induced gene expression, secretion and proteolytic activity of matrix metalloproteinase in cultured articular chondrocytes and production of matrix metalloproteinase in the knee joint of rat
- Luteolin Inhibits the Activity, Secretion and Gene Expression of MMP-3 in Cultured Articular Chondrocytes and Production of MMP-3 in the Rat Knee