Radioprotective effects of fucoidan on bone marrow cells: improvement of the cell survival and immunoreactivity
- Affiliations
-
- 1College of Veterinary Medicine, Cheju National University, Jeju 690-756, Korea. jooh@cheju.ac.kr
- 2Department of Pharmacology, College of Medicine, Cheju National University, Jeju 690-756, Korea.
- 3Applied Radiological Science Research Institute, Cheju National University, Jeju 690-756, Korea.
- 4The Research Institute for Subtropical Agriculture and Biotechnology, Cheju National University, Jeju 690-756, Korea.
- KMID: 1104910
- DOI: http://doi.org/10.4142/jvs.2008.9.4.359
Abstract
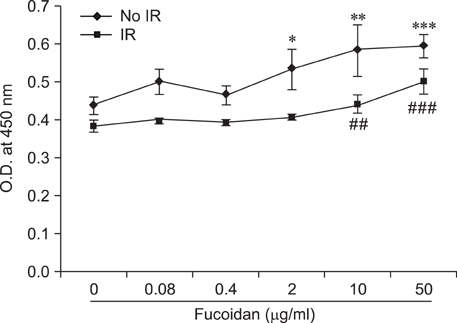
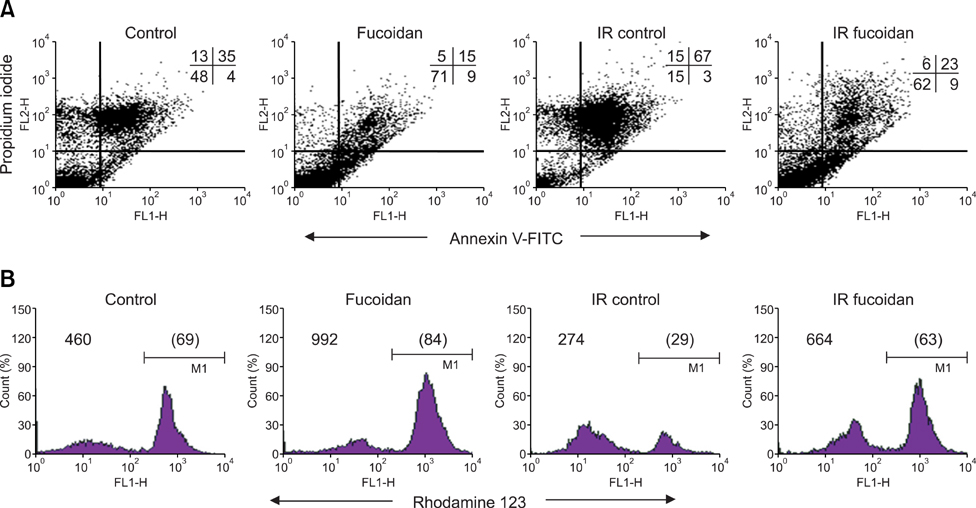
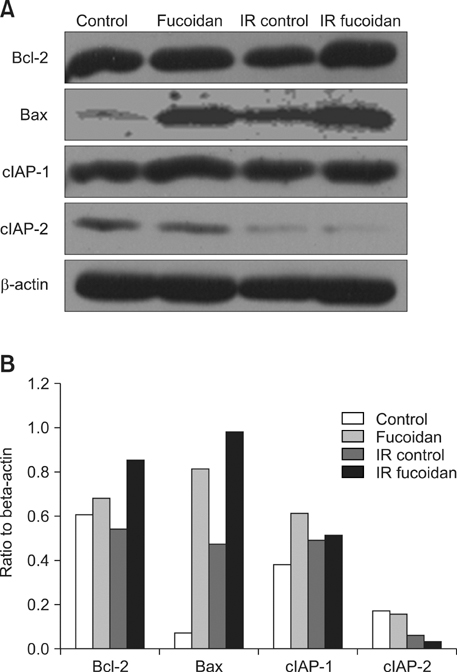
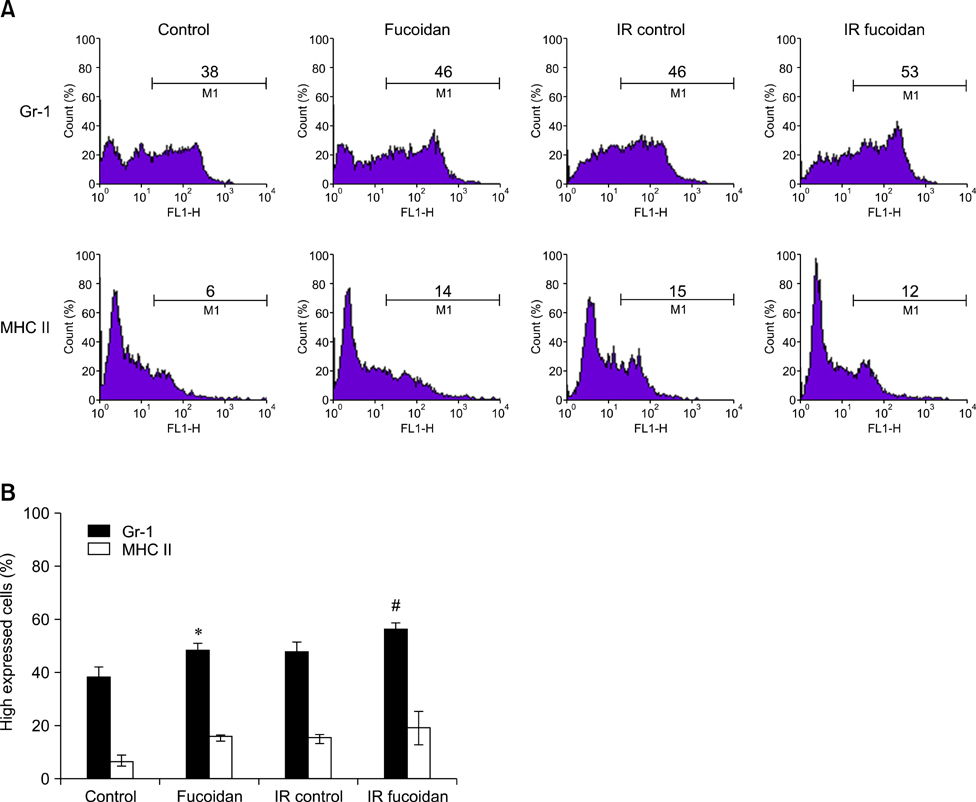
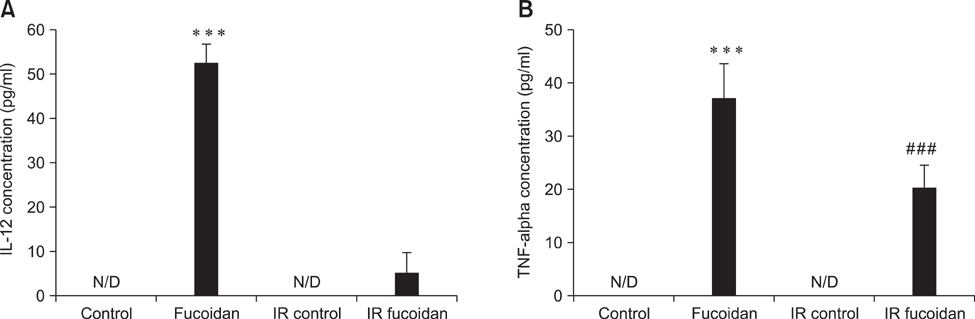
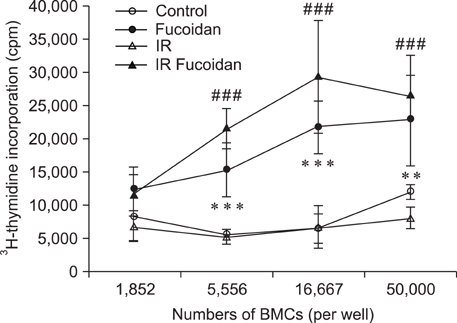
- Fucoidan is a sulfated polysaccharide purified from brown algae including Fucus vesiculosus and has a variety of biological effects including mobilization of hematopoietic progenitor cells. Recently, we demonstrated that fucoidan stimulates the antigen-presenting functions of dendritic cells. In this study, we investigated the radioprotective effects of fucoidan on bone marrow cells (BMCs), which are the main cellular reservoir for the hematopoietic and immune system. To evaluate the effects of fucoidan, we assayed cell viability and immune responses. In a viability assay, fucoidan significantly increased the viability of BMCs. Based on the results of flow cytometric analysis, the increased viability of fucoidan-treated BMCs was attributed to the inhibition of radiation-induced apoptosis. Furthermore, fucoidan altered the production of immune-related cytokines from BMCs and increased the capability of BMCs to induce proliferation of allogeneic splenocytes. Taken together, our study demonstrated that fucoidan has radioprotective effects on BMCs with respect to cell viability and immunoreactivity. These results may provide valuable information, useful in the field of radiotherapy.
Keyword
MeSH Terms
-
Animals
Bone Marrow Cells/*drug effects/*radiation effects
Cell Death/drug effects/radiation effects
Cell Proliferation
Cell Survival/drug effects
Cells, Cultured
Female
Gamma Rays/*adverse effects
Mice
Mice, Inbred BALB C
Mice, Inbred C57BL
Polysaccharides/*pharmacology
Radiation-Protective Agents/*pharmacology
Spleen/cytology
Figure
Cited by 4 articles
-
Paclitaxel inhibits the hyper-activation of spleen cells by lipopolysaccharide and induces cell death
Hyun-Ji Kim, Hong-Gu Joo
J Vet Sci. 2016;17(4):453-458. doi: 10.4142/jvs.2016.17.4.453.Immunostimulatory Effects of β-glucan Purified from
Paenibacillus polymyxa JB115 on Mouse Splenocytes
Ji-Mi Kim, Hong-Gu Joo
Korean J Physiol Pharmacol. 2012;16(4):225-230. doi: 10.4196/kjpp.2012.16.4.225.Stimulatory Effects of Ginsan on the Proliferation and Viability of Mouse Spleen Cells
Eun-Ju Ko, Hong-Gu Joo
Korean J Physiol Pharmacol. 2010;14(3):133-137. doi: 10.4196/kjpp.2010.14.3.133.Fucoidan Enhances the Survival and Sustains the Number of Splenic Dendritic Cells in Mouse Endotoxemia
Eun-Ju Ko, Hong-Gu Joo
Korean J Physiol Pharmacol. 2011;15(2):89-94. doi: 10.4196/kjpp.2011.15.2.89.
Reference
-
1. Arora R, Gupta D, Chawla R, Sagar R, Sharma A, Kumar R, Prasad J, Singh S, Samanta N, Sharma RK. Radioprotection by plant products: present status and future prospects. Phytother Res. 2005. 19:1–22.
Article2. Choi EM, Kim AJ, Kim YO, Hwang JK. Immunomodulating activity of arabinogalactan and fucoidan in vitro. J Med Food. 2005. 8:446–453.3. Frenette PS, Weiss L. Sulfated glycans induce rapid hematopoietic progenitor cell mobilization: evidence for selectin-dependent and independent mechanisms. Blood. 2000. 96:2460–2468.
Article4. Gross A, McDonnell JM, Korsmeyer SJ. BCL-2 family members and the mitochondria in apoptosis. Genes Dev. 1999. 13:1899–1911.
Article5. Hosseinimehr SJ. Trends in the development of radioprotective agents. Drug Discov Today. 2007. 12:794–805.
Article6. Irhimeh MR, Fitton JH, Lowenthal RM. Fucoidan ingestion increases the expression of CXCR4 on human CD34+ cells. Exp Hematol. 2007. 35:989–994.
Article7. Ivanova T, Han Y, Son HJ, Yun YS, Song JY. Antimutagenic effect of polysaccharide ginsan extracted from Panax ginseng. Food Chem Toxicol. 2006. 44:517–521.
Article8. Joo HG, Goedegebuure PS, Sadanaga N, Nagoshi M, von Bernstorff W, Eberlein TJ. Expression and function of galectin-3, a β-galactoside-binding protein in activated T lymphocytes. J Leukoc Biol. 2001. 69:555–564.9. Kim HJ, Kim MH, Byon YY, Park JW, Jee Y, Joo HG. Radioprotective effects of an acidic polysaccharide of Panax ginseng on bone marrow cells. J Vet Sci. 2007. 8:39–44.
Article10. Kim MH, Joo HG. Immunostimulatory effects of fucoidan on bone marrow-derived dendritic cells. Immunol Lett. 2008. 115:138–143.
Article11. Lake AC, Vassy R, Di Benedetto M, Lavigne D, Le Visage C, Perret GY, Letourneur D. Low molecular weight fucoidan increases VEGF165-induced endothelial cell migration by enhancing VEGF165 binding to VEGFR-2 and NRP1. J Biol Chem. 2006. 281:37844–37852.
Article12. Liston P, Fong WG, Korneluk RG. The inhibitors of apoptosis: there is more to life than Bcl2. Oncogene. 2003. 22:8568–8580.
Article13. Maruyama H, Tamauchi H, Iizuka M, Nakano T. The role of NK cells in antitumor activity of dietary fucoidan from Undaria pinnatifida sporophylls (Mekabu). Planta Med. 2006. 72:1415–1417.
Article14. Opferman JT, Korsmeyer SJ. Apoptosis in the development and maintenance of the immune system. Nat Immunol. 2003. 4:410–415.
Article15. Rades D, Fehlauer F, Bajrovic A, Mahlmann B, Richter E, Alberti W. Serious adverse effects of amifostine during radiotherapy in head and neck cancer patients. Radiother Oncol. 2004. 70:261–264.
Article16. Rothe M, Pan MG, Henzel WJ, Ayres TM, Goeddel DV. The TNFR2-TRAF signaling complex contains two novel proteins related to baculoviral inhibitor of apoptosis proteins. Cell. 1995. 83:1243–1252.
Article17. Santini V, Giles FJ. The potential of amifostine: from cytoprotectant to therapeutic agent. Haematologica. 1999. 84:1035–1042.18. Subramanian M, Chintalwar GJ, Chattopadhyay S. Antioxidant and radioprotective properties of an Ocimum sanctum polysaccharide. Redox Rep. 2005. 10:257–264.
Article19. Sweeney EA, Lortat-Jacob H, Priestley GV, Nakamoto B, Papayannopoulou T. Sulfated polysaccharides increase plasma levels of SDF-1 in monkeys and mice: involvement in mobilization of stem/progenitor cells. Blood. 2002. 99:44–51.
Article20. Uren AG, Pakusch M, Hawkins CJ, Puls KL, Vaux DL. Cloning and expression of apoptosis inhibitory protein homologs that function to inhibit apoptosis and/or bind tumor necrosis factor receptor-associated factors. Proc Natl Acad Sci USA. 1996. 93:4974–4978.
Article21. Wang ZW, Zhou JM, Huang ZS, Yang AP, Liu ZC, Xia YF, Zeng YX, Zhu XF. Aloe polysaccharides mediated radioprotective effect through the inhibition of apoptosis. J Radiat Res. 2004. 45:447–454.
Article22. Wrembel-Wargocka J, Jabłońska H, Chomiczewski K. Clinical use of amifostine (WR-2721) as a preparation protecting healthy tissues from the cytotoxic effects of chemotherapy and radiation therapy. Przegl Lek. 1996. 53:820–825.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Fucoidan Enhances the Survival and Sustains the Number of Splenic Dendritic Cells in Mouse Endotoxemia
- Radioprotective effects of an acidic polysaccharide of Panax ginseng on bone marrow cells
- Antimetastatic effect of fucoidan against non-small cell lung cancer by suppressing non-receptor tyrosine kinase and extracellular signalrelated kinase pathway
- Fucoidan promotes osteoblast differentiation via JNK- and ERK-dependent BMP2-Smad 1/5/8 signaling in human mesenchymal stem cells
- Antitumor Effects of Fucoidan on Human Colon Cancer Cells via Activation of Akt Signaling